Mastering the Cheng-Prusoff Equation: A Practical Guide to Converting IC50 to Ki in Drug Discovery
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on the application, interpretation, and validation of the Cheng-Prusoff equation for converting experimentally determined IC50 values to...
Mastering the Cheng-Prusoff Equation: A Practical Guide to Converting IC50 to Ki in Drug Discovery
Abstract
This article provides a comprehensive guide for researchers, scientists, and drug development professionals on the application, interpretation, and validation of the Cheng-Prusoff equation for converting experimentally determined IC50 values to inhibition constants (Ki). We explore the biochemical and mathematical foundations of competitive binding, deliver step-by-step methodological workflows for accurate calculation, address common pitfalls and optimization strategies, and compare the Cheng-Prusoff approach with modern computational and direct assay methods. The goal is to empower practitioners with the knowledge to correctly derive Ki values, critical for comparing compound potency, informing SAR, and selecting candidates for preclinical development.
Understanding the Cheng-Prusoff Equation: The Science of IC50 and Ki
Introduction In the quantitative analysis of drug-receptor interactions, the terms IC50 and Ki are fundamental, yet they are often erroneously used interchangeably. This distinction is paramount in pharmacology and drug discovery. IC50 (half-maximal inhibitory concentration) is an empirical, assay-dependent measure of a compound's potency. In contrast, Ki (inhibition constant) is an absolute, assay-independent thermodynamic constant describing the affinity of an inhibitor for its target. This application note, framed within the context of research on the Cheng-Prusoff equation for converting IC50 to Ki, details the experimental protocols and analytical frameworks necessary to correctly determine and interpret these parameters.
Quantitative Data Summary
Table 1: Core Definitions and Properties of IC50 vs. Ki
| Parameter | Definition | Units | Assay-Dependent? | Reflects | Determined From |
|---|---|---|---|---|---|
| IC50 | Concentration of inhibitor required to reduce a measured biological response/activity by 50%. | Molar (e.g., nM, µM) | Yes (varies with assay conditions) | Functional Potency | Directly from dose-response curves. |
| Ki | Equilibrium dissociation constant for the inhibitor-target complex. | Molar (e.g., nM, µM) | No (true constant for a given target & condition) | Binding Affinity | Derived from IC50 using Cheng-Prusoff or direct binding assays. |
Table 2: Key Variables in the Cheng-Prusoff Equation and Their Impact
| Variable | Symbol | Description | Impact on Ki Calculation |
|---|---|---|---|
| Substrate Concentration | [S] | Concentration of the varied substrate in the assay. | Higher [S] requires higher [I] for inhibition, inflating IC50. |
| Michaelis Constant | Km | Substrate concentration at half Vmax. | Determines the sensitivity of the enzyme to the substrate. |
| Ligand Concentration | [L] | Concentration of radiolabeled ligand in binding assays. | Higher [L] inflates observed IC50. |
| Ligand Dissociation Constant | Kd | Equilibrium dissociation constant of the radiolabeled ligand. | A fixed property of the probe ligand. |
| Inhibition Mode | - | Competitive, non-competitive, uncompetitive, etc. | Determines the correct form of the Cheng-Prusoff equation to use. |
Experimental Protocols
Protocol 1: Determination of IC50 via Enzymatic Activity Assay Objective: To generate a dose-response curve for an enzyme inhibitor and determine the experimental IC50 value.
- Reagent Preparation: Prepare a serial dilution (e.g., 10 concentrations, 3-fold dilutions) of the test inhibitor in assay buffer (containing DMSO ≤1%). Prepare enzyme, substrate, and cofactor solutions per manufacturer specifications.
- Assay Assembly: In a 96-well plate, combine assay buffer, enzyme, and inhibitor (or vehicle control). Pre-incubate for 15-30 minutes at assay temperature to allow inhibitor binding.
- Reaction Initiation: Initiate the enzymatic reaction by adding the substrate at its predetermined Km concentration (for competitive inhibitors). Include no-enzyme (background) and no-inhibitor (positive control) wells.
- Signal Measurement: Monitor product formation kinetically using a plate reader (absorbance, fluorescence, or luminescence) for 15-60 minutes, ensuring linear reaction kinetics.
- Data Analysis: Calculate reaction velocity (V) for each well. Normalize data: % Activity = (Vinhibitor / Vpositive_control) * 100. Fit normalized data to a 4-parameter logistic (sigmoidal) model: Y = Bottom + (Top-Bottom) / (1 + 10^((LogIC50 - X)*HillSlope)). The IC50 is the concentration at Y=50%.
Protocol 2: Conversion of IC50 to Ki Using the Cheng-Prusoff Equation (Competitive Inhibition) Objective: To calculate the absolute affinity constant (Ki) from an IC50 determined under defined assay conditions.
- Prerequisite Data: Obtain the experimental IC50 from Protocol 1. Precisely know the concentration of the varied substrate ([S]) used in the assay and the Michaelis constant (Km) for that substrate under identical conditions. Km must be determined in a separate experiment using Michaelis-Menten kinetics.
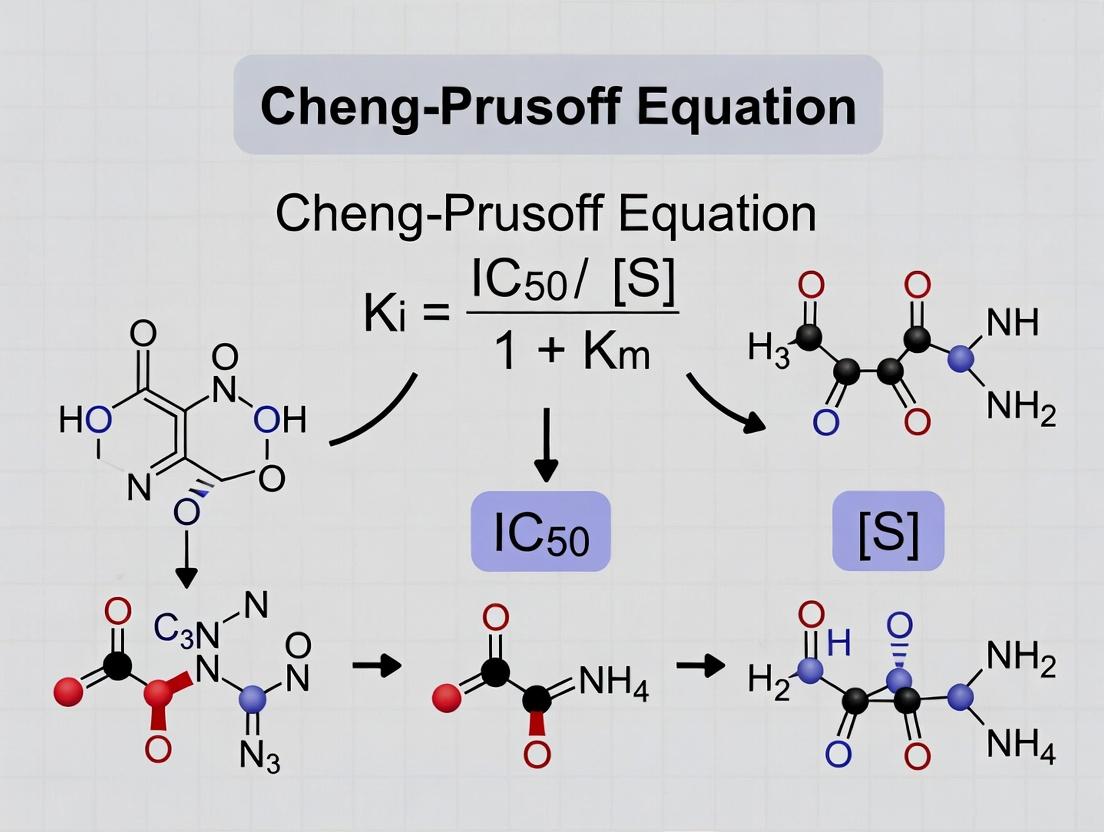
- Equation Application: Apply the Cheng-Prusoff equation for competitive enzyme inhibition: Ki = IC50 / (1 + [S]/Km).
- Calculation: For example, if IC50 = 100 nM, [S] = Km = 10 µM, then Ki = 100 nM / (1 + 10/10) = 50 nM.
- Critical Validation: Verify the assumption of competitive inhibition (e.g., via Lineweaver-Burk plots). For non-competitive inhibition: Ki = IC50. For binding assays (e.g., radioligand competition): Ki = IC50 / (1 + [L]/Kd), where [L] is free radioligand concentration and Kd is its dissociation constant.
Visualizations
IC50 to Ki Conversion Workflow
Competitive Inhibition Binding Pathway
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for IC50/Ki Determination
| Reagent/Material | Function & Importance |
|---|---|
| Recombinant Target Protein (Enzyme/Receptor) | The purified biological target. High purity and activity are critical for reproducible kinetics. |
| Fluorogenic/Chemiluminescent Substrate | Allows sensitive, continuous monitoring of enzyme velocity without stopping the reaction, ideal for IC50 curves. |
| Radioactive Ligand (e.g., ³H, ¹²⁵I-labeled) | For direct binding/competition assays to determine Kd and Ki without assumptions about inhibition mechanism. |
| Positive Control Inhibitor (Known Ki) | Validates assay performance and serves as a benchmark for comparing newly derived IC50/Ki values. |
| Low-Binding Microplates & Tips | Minimizes nonspecific adsorption of inhibitor/target, especially critical for hydrophobic compounds. |
| Dispensing Robot/Liquid Handler | Ensures precision and reproducibility when dispensing serial dilutions of inhibitors and reagents. |
| Data Analysis Software (e.g., Prism, SigmaPlot) | For robust nonlinear regression fitting of dose-response and kinetic data to derive IC50, Km, and Ki. |
Application Notes
The Cheng-Prusoff equation, first published in 1973, provides the essential mathematical relationship for converting experimentally measured half-maximal inhibitory concentration (IC₅₀) values to the absolute inhibition constant (Kᵢ). This conversion is fundamental to modern pharmacology and drug discovery, allowing for the accurate comparison of ligand affinity across different experimental conditions. Its derivation is based on the principles of competitive inhibition within Michaelis-Menten and receptor-ligand binding kinetics.
The core equations are:
- For enzyme assays: Kᵢ = IC₅₀ / (1 + [S]/Kₘ)
- For receptor binding assays: Kᵢ = IC₅₀ / (1 + [L]/Kₐ)
Where [S] is substrate concentration, Kₘ is the Michaelis constant, [L] is the concentration of the radioligand, and Kₐ is its dissociation constant.
Critical Assumptions & Limitations:
- The inhibitor must act competitively.
- The system must be at equilibrium.
- Binding follows the law of mass action.
- There is no cooperativity or allosteric modulation.
- The concentration of the inhibitor is much greater than that of the enzyme/receptor ([I] >> [E]), ensuring free inhibitor concentration is approximated by total added concentration.
Failure to meet these assumptions, particularly in complex systems like cell-based functional assays, can lead to significant inaccuracies. Contemporary research emphasizes rigorous experimental design and validation to ensure the correct application of the Cheng-Prusoff relationship.
Table 1: Impact of Substrate/Ligand Concentration on IC₅₀ to Kᵢ Conversion
| [S] or [L] Concentration | Relation to Kₘ or Kₐ | IC₅₀ to Kᵢ Ratio | Practical Implication |
|---|---|---|---|
| [S] = 0.1 * Kₘ[L] = 0.1 * Kₐ | 10-fold below | IC₅₀ ≈ 1.1 * Kᵢ | IC₅₀ is a good approximation of Kᵢ. |
| [S] = Kₘ[L] = Kₐ | At the constant | IC₅₀ = 2 * Kᵢ | IC₅₀ overestimates Kᵢ by 2-fold. |
| [S] = 10 * Kₘ[L] = 10 * Kₐ | 10-fold above | IC₅₀ ≈ 11 * Kᵢ | IC₅₀ significantly overestimates Kᵢ; conversion is mandatory. |
Table 2: Common Experimental Errors Affecting Cheng-Prusoff Validity
| Error Type | Effect on IC₅₀ | Effect on Calculated Kᵢ | Recommended Mitigation |
|---|---|---|---|
| Non-competitive inhibition | Altered | Invalid | Use appropriate model (e.g., non-competitive fit). |
| Substrate depletion | Overestimated | Overestimated | Ensure [S] >> [E]; use kinetic assays. |
| Ligand depletion | Overestimated | Overestimated | Use low receptor concentration (<0.1 * Kₐ). |
| Incorrect Kₘ/Kₐ value | N/A | Propagated error | Determine Kₘ/Kₐ precisely under identical conditions. |
Experimental Protocols
Protocol 1: Determining Kᵢ for a Competitive Enzyme Inhibitor
Objective: To determine the absolute inhibition constant (Kᵢ) of a novel compound for a purified enzyme using a spectrophotometric activity assay.
Materials: See "The Scientist's Toolkit" below. Procedure:
- Determine Kₘ: Perform a Michaelis-Menten experiment.
- Prepare a dilution series of the substrate (e.g., 0.2Kₘ to 5Kₘ, 8 concentrations) in assay buffer.
- In a 96-well plate, mix 70 µL substrate solution with 20 µL assay buffer. Initiate reaction by adding 10 µL of enzyme solution.
- Immediately monitor product formation spectrophotometrically for 5-10 minutes.
- Fit initial velocity (v₀) vs. [S] data to the Michaelis-Menten equation using non-linear regression to obtain Kₘ.
Determine IC₅₀:
- Prepare a serial dilution of the inhibitor (e.g., 10 concentrations across a 3-log range) in DMSO, keeping final DMSO ≤1%.
- In a plate, combine 60 µL of a fixed substrate concentration ([S], typically ≈ Kₘ), 20 µL of inhibitor dilution (or vehicle control), and 10 µL of assay buffer. Pre-incubate for 15 min.
- Initiate reaction with 10 µL of enzyme solution. Measure initial velocity as in Step 1.
- Plot % activity (relative to vehicle control) vs. log[Inhibitor]. Fit data to a 4-parameter logistic (sigmoidal) model to obtain IC₅₀.
Calculate Kᵢ: Apply the Cheng-Prusoff equation: Kᵢ = IC₅₀ / (1 + [S]/Kₘ).
Protocol 2: Determining Kᵢ in a Radioligand Receptor Binding Assay
Objective: To determine the inhibition constant (Kᵢ) of an unlabeled compound competing with a radioligand for a membrane-bound receptor.
Materials: See "The Scientist's Toolkit" below. Procedure:
- Determine Kₐ (Kd) of Radioligand: Perform a saturation binding experiment.
- Prepare a dilution series of the radioligand (e.g., 0.1Kₐ to 10Kₐ, 10 concentrations) in binding buffer.
- In a deep-well plate, combine 400 µL membrane preparation, 50 µL radioligand dilution, and 50 µL buffer (for total binding) or excess cold ligand (for nonspecific binding). Perform in triplicate.
- Incubate to equilibrium (time/temp determined from kinetics experiments). Terminate by rapid filtration through GF/B filters. Wash filters, dry, and count radioactivity.
- Plot specific binding (Total - Nonspecific) vs. [Radioligand]. Fit data to a one-site specific binding model to obtain Kₐ (Kd).
Determine IC₅₀ of Competitor:
- Prepare a serial dilution of the unlabeled test compound (e.g., 10 concentrations) in buffer/DMSO.
- In a plate, combine 350 µL membrane prep, 50 µL of a fixed concentration of radioligand ([L], typically ≈ 0.5-1 * Kₐ), and 50 µL of test compound dilution (or vehicle). Include total and nonspecific binding controls.
- Incubate, filter, wash, and count as in Step 1.
- Plot % specific binding (relative to total binding control) vs. log[Competitor]. Fit sigmoidal curve to obtain IC₅₀.
Calculate Kᵢ: Apply the Cheng-Prusoff equation: Kᵢ = IC₅₀ / (1 + [L]/Kₐ).
Visualizations
Title: Decision Workflow for Converting IC50 to Ki
Title: Relationship Between Experimental Data and Ki Calculation
The Scientist's Toolkit
Table 3: Essential Reagents and Materials for Cheng-Prusoff-Based Studies
| Item | Function & Relevance | Example/Note |
|---|---|---|
| Purified Target Enzyme / Membrane Prep | Source of the biological target. Purity and activity directly impact Kₘ and IC₅₀ accuracy. | Recombinant enzyme, cell/ tissue membrane fractions. |
| Validated Substrate or Radioligand | The molecule whose turnover or displacement is measured. Its Kₘ/Kₐ must be precisely known. | Chromogenic/fluorogenic substrate; [³H]-, [¹²⁵I]-labeled ligands. |
| Test Inhibitor Compounds | The molecules being characterized. Requires accurate serial dilution in compatible solvent. | High-purity small molecules, solubilized in DMSO. |
| Assay Buffer System | Maintains pH, ionic strength, and cofactors to ensure proper enzyme/receptor function. | Often includes Tris or HEPES, salts, Mg²⁺, BSA, protease inhibitors. |
| Multi-well Plates & Plate Reader | High-throughput format for running dose-response curves under identical conditions. | 96- or 384-well plates; spectrophotometer or fluorimeter. |
| Filtration Harvester & GF/B Filters | Essential for separating bound from free radioligand in receptor binding assays. | Brand-specific systems (e.g., Brandel, PerkinElmer). |
| Scintillation Counter / Beta Counter | Quantifies radioactivity from filter-bound radioligand to determine binding levels. | Requires appropriate scintillation cocktail. |
| Non-linear Regression Software | Fits experimental data to kinetic and binding models to derive IC₅₀, Kₘ, and Kₐ. | GraphPad Prism, SigmaPlot, specialized commercial packages. |
Application Notes and Protocols
Within the broader thesis on the Cheng-Prusoff equation for converting IC₅₀ to Kᵢ values, this document provides a rigorous dissection of its mathematical derivation and the critical assumptions underlying its application in competitive binding assays. Accurate Kᵢ determination is paramount for researchers and drug development professionals to compare ligand affinities across different experimental conditions.
Mathematical Derivation from Mass-Action Kinetics
The Cheng-Prusoff equation is derived from the fundamental principles of competitive inhibition for a simple one-site receptor-ligand interaction at equilibrium. The core components are:
- [L]: Concentration of the free radioligand.
- [I]: Concentration of the free inhibitor.
- K_d: Dissociation constant of the radioligand.
- K_i: Dissociation constant of the inhibitor (the desired parameter).
- [RL]: Concentration of the receptor-ligand complex.
The derivation begins with the standard equation for fractional occupancy of the receptor by the radioligand in the presence of a competitive inhibitor:
Fractional Occupancy = [RL] / [Rtotal] = [L] / ( [L] + Kd (1 + [I]/K_i) )
The IC₅₀ is defined as the concentration of inhibitor that reduces the specific binding of the radioligand by 50%. At this point:
- Binding in absence of inhibitor: [RL]₀ = [L] / ([L] + K_d)
- Binding in presence of IC₅₀: [RL]_IC = 0.5 * [RL]₀
Substituting into the fractional occupancy equation and solving for IC₅₀ yields the classic Cheng-Prusoff relationship:
IC₅₀ = Ki * ( [L] / Kd + 1 )
The more common rearranged form for calculating Kᵢ is:
Ki = IC₅₀ / ( 1 + [L] / Kd )
This derivation leads directly to the core quantitative relationship summarized in Table 1.
Table 1: Core Cheng-Prusoff Equation Variables
| Variable | Description | Unit | Experimental Determination |
|---|---|---|---|
| IC₅₀ | Inhibitor concentration at 50% radioligand binding reduction | M (e.g., nM) | From dose-response curve (Protocol 1) |
| [L] | Free concentration of the competing radioligand | M | Typically the added concentration; see Assumption 1. |
| K_d | Dissociation constant of the radioligand-receptor complex | M | From saturation binding isotherm (Protocol 2) |
| K_i | Dissociation constant of the inhibitor-receptor complex | M | Calculated output. |
Critical Assumptions and Their Implications
The validity of the Kᵢ calculation is contingent upon strict adherence to several assumptions. Deviations can lead to significant systematic errors.
Table 2: Key Assumptions of the Cheng-Prusoff Equation
| Assumption | Implication | Consequence of Violation |
|---|---|---|
| 1. Equilibrium Conditions | The reaction system (R + L + I) is at steady-state. | Time-dependent assays yield inaccurate IC₅₀. |
| 2. Competitive Inhibition | Inhibitor and radioligand bind to the same site on the receptor in a mutually exclusive manner. | Overestimates affinity for allosteric or non-competitive inhibitors. |
| 3. Law of Mass Action | Simple 1:1 binding with no cooperativity. | Complex binding kinetics invalidate the simple model. |
| 4. [L] >> [R_total] | Free radioligand concentration approximates total added. Prevents ligand depletion. | Significant ligand depletion (>10%) leads to underestimation of Kᵢ. |
| 5. [I] >> [R_total] | Free inhibitor concentration approximates total added. Prevents inhibitor depletion. | Significant inhibitor depletion leads to overestimation of IC₅₀ and Kᵢ. |
| 6. No Ligand/Inhibitor Binding to Other Sites | The system contains only the target receptor. | Nonspecific binding or off-target effects skew the dose-response. |
Experimental Protocols
Protocol 1: Determining IC₅₀ via Competitive Binding Assay
- Objective: To generate a dose-response curve for the inhibitor.
- Procedure:
- Prepare a constant concentration of receptor preparation (membrane homogenate, whole cells) in assay buffer.
- Add a constant, near-Kd concentration of radioligand ([L] ≈ Kd, see Table 1).
- Add a serial dilution of the inhibitor (typically spanning 10⁻¹² to 10⁻⁵ M, 12 points in duplicate).
- Include controls for total binding (radioligand + receptor) and nonspecific binding (NSB, radioligand + receptor + excess cold ligand).
- Incubate to equilibrium (time determined empirically, typically 60-120 min at assay temperature).
- Separate bound from free radioligand (via filtration, centrifugation, or SPA bead settling).
- Quantify bound radioactivity (scintillation counting, gamma counter).
- Fit normalized specific binding data (% of total binding minus NSB) vs. log[Inhibitor] to a four-parameter logistic (4PL) model to obtain IC₅₀.
Protocol 2: Determining K_d via in-parallel Saturation Binding
- Objective: To determine the radioligand's K_d under identical assay conditions.
- Procedure:
- Using the same receptor preparation and buffer as Protocol 1.
- Perform a saturation binding isotherm: incubate receptor with increasing concentrations of radioligand (e.g., 0.1x to 10x estimated K_d, 12 points).
- At each [L], measure total binding and nonspecific binding (with excess cold ligand).
- Calculate specific binding = Total - NSB.
- Plot specific bound vs. [L] and fit data directly to the one-site specific binding model:
Y = B_max * X / (K_d + X)to derive Kd and Bmax (receptor density).
Visualizing the Framework and Workflow
Cheng-Prusoff Conceptual Dependency Diagram
K_i Determination Experimental Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Competitive Binding Assays
| Item | Function & Critical Consideration |
|---|---|
| Purified Receptor Preparation (e.g., membrane fractions, whole cells) | Source of the target protein. Consistency in preparation across K_d and IC₅₀ experiments is vital. |
| Tritiated (³H) or Iodinated (¹²⁵I) Radioligand | High-affinity, high-specific-activity tracer. Must have known pharmacological profile for the target. |
| Unlabeled ("Cold") Competitor/Inhibitor | The compound under investigation. Purity and accurate concentration are critical. |
| Unlabeled Ligand for NSB Definition (e.g., isoproterenol for β-adrenoceptors) | Used at high excess (100-1000x K_d) to define nonspecific binding. Should be highly selective. |
| Assay Buffer with Protease Inhibitors | Maintains pH, ionic strength, and protein stability. May require specific ions (e.g., Mg²⁺ for GPCRs). |
| Filtration Apparatus (GF/B or GF/C filters, harvester) or SPA Beads | For rapid separation of bound from free radioligand. Filter material must minimize nonspecific ligand binding. |
| Scintillation Counter / Gamma Counter | For quantifying bound radioactivity. Requires appropriate scintillation cocktail for filters. |
| Nonlinear Curve-Fitting Software (e.g., Prism, GraphPad) | To fit data to 4PL (IC₅₀) and one-site binding (K_d) models. Weighting and model selection are important. |
The accurate determination of the inhibition constant (Kᵢ) is a cornerstone of quantitative enzymology and drug discovery. While the half-maximal inhibitory concentration (IC₅₀) is an empirical, experiment-derived value, it is dependent on assay conditions such as substrate concentration. The Cheng-Prusoff equation suite provides the critical framework for converting IC₅₀ to the absolute affinity metric Kᵢ, but its correct application is wholly dependent on the accurate mechanistic diagnosis of the inhibition modality—Competitive, Non-Competitive, or Uncompetitive. Misidentification leads to significant errors in Kᵢ estimation, potentially derailing structure-activity relationships and lead optimization. This application note details the core equation forms, their derivations, and provides robust experimental protocols for their validation, serving as an essential practical guide for research aligned with advancing the precision of Cheng-Prusoff methodologies.
Core Equation Forms and Theory
The mode of inhibition defines the interaction between enzyme (E), substrate (S), and inhibitor (I). Each modality has a distinct effect on the Michaelis-Menten parameters (Kₘ and Vₘₐₓ) and, consequently, a unique form of the Cheng-Prusoff relationship.
Table 1: Summary of Inhibition Modalities, Effects, and Core Cheng-Prusoff Equations
| Inhibition Type | Mechanism (Binding Site) | Effect on Kₘ (Apparent) | Effect on Vₘₐₓ (Apparent) | Core Rate Equation (vᵢ) | Cheng-Prusoff Conversion (IC₅₀ to Kᵢ) |
|---|---|---|---|---|---|
| Competitive | Binds active site, competes with S. | Increases | Unchanged | vᵢ = (Vₘₐₓ [S]) / (Kₘ(1 + [I]/Kᵢ) + [S]) | Kᵢ = IC₅₀ / (1 + [S]/Kₘ) |
| Non-Competitive | Binds allosteric site, affects catalysis. | Unchanged | Decreases | vᵢ = (Vₘₐₓ [S]) / ((Kₘ + [S])(1 + [I]/Kᵢ)) | Kᵢ = IC₅₀ |
| Uncompetitive | Binds only enzyme-substrate complex (ES). | Decreases | Decreases | vᵢ = (Vₘₐₓ [S]) / (Kₘ + S) | Kᵢ = IC₅₀ / (1 + [S]/Kₘ) |
Note: For Non-Competitive inhibition, this assumes pure, linear non-competitive inhibition where *Kᵢ = Kᵢ' (the binding constant for EI and EIS complexes are equal). [S] is the fixed substrate concentration used in the IC₅₀ assay.*
Experimental Protocols for Diagnosing Inhibition Modality
A definitive diagnosis requires evaluating the effect of the inhibitor on enzyme kinetics under varied substrate concentrations.
Protocol 1: Comprehensive Steady-State Kinetics Assay for Modality Diagnosis
Objective: To determine the inhibition modality and extract Kᵢ via global fitting of kinetic data.
Materials: See "Scientist's Toolkit" below.
Procedure:
- Enzyme Preparation: Dilute purified enzyme in assay buffer to a working concentration within the linear reaction range. Keep on ice.
- Substrate & Inhibitor Plating:
- Prepare a serial dilution of the test inhibitor (e.g., 8 concentrations covering 0.1x to 100x the expected Kᵢ) in DMSO, then in assay buffer (final DMSO ≤1%).
- Prepare 4-5 different substrate concentrations bracketing the known Kₘ (e.g., 0.25Kₘ, 0.5Kₘ, 1Kₘ, 2Kₘ, 4Kₘ).
- In a 96-well plate, add substrate solutions (in triplicate). Add inhibitor dilutions to create a matrix where every [S] is tested against every [I]. Include control wells with no inhibitor (100% activity) and no enzyme (background).
- Reaction Initiation & Measurement:
- Start reactions by adding a fixed volume of enzyme solution using a multi-channel pipette.
- Immediately place the plate in a pre-warmed microplate reader.
- Monitor the increase in product (e.g., absorbance, fluorescence) kinetically for 5-15 minutes.
- Data Analysis:
- Calculate initial velocities (v₀) from the linear portion of progress curves.
- For each substrate concentration, plot v₀ vs. [I] to generate IC₅₀ curves.
- Global Nonlinear Regression: Fit the complete dataset (all v₀, [S], [I]) simultaneously to the full competitive, non-competitive, and uncompetitive rate equations using software (e.g., GraphPad Prism, SigmaPlot).
- The model with the lowest sum-of-squares, smallest standard errors for fitted parameters (Kₘ, Vₘₐₓ, Kᵢ), and most random residuals is the correct mechanistic model.
- Visualize the diagnosis with double-reciprocal (Lineweaver-Burk) plots.
Protocol 2: Rapid IC₅₀ Shift Assay for Preliminary Classification
Objective: A faster, qualitative method to distinguish competitive from non/uncompetitive patterns.
Procedure:
- Perform two separate IC₅₀ determinations as per standard protocols.
- Condition A: Use substrate concentration at [S] = Kₘ.
- Condition B: Use a high substrate concentration, [S] = 5-10 x Kₘ.
- Fit dose-response curves (v vs. log[I]) for both conditions to a 4-parameter logistic model to obtain IC₅₀(ᴬ) and IC₅₀(ᴮ).
- Interpretation:
- If IC₅₀(ᴮ) >> IC₅₀(ᴬ) (significant right-shift), inhibition is Competitive.
- If IC₅₀ is largely unchanged, inhibition is Non-Competitive.
- If IC₅₀(ᴬ) > IC₅₀(ᴮ) (left-shift), inhibition is likely Uncompetitive.
Visualization of Mechanistic Pathways and Workflows
Title: Enzyme Inhibition Mechanistic Pathways
Title: Experimental Workflow for Inhibition Modality Diagnosis
The Scientist's Toolkit
Table 2: Key Research Reagent Solutions for Inhibition Studies
| Reagent / Material | Function & Importance in Cheng-Prusoff Context |
|---|---|
| High-Purity Recombinant Enzyme | Essential for obtaining reliable, reproducible kinetic parameters (Kₘ, k_cat) free from interfering contaminants. |
| Kinetically Validated Substrate | Must generate a stable, measurable signal with known Kₘ under assay conditions. Critical for setting correct [S] levels. |
| DMSO (Chromatography Grade) | Universal solvent for compound libraries. Must be used at consistent, low concentration (≤1%) to avoid enzyme denaturation. |
| Assay Buffer with Cofactors | Optimized pH, ionic strength, and containing essential cofactors (Mg²⁺, ATP, etc.) to maintain native enzyme activity. |
| Positive Control Inhibitor | A well-characterized inhibitor of known modality and Kᵢ for the target or a related enzyme. Serves as an assay validation standard. |
| Detection Reagents (e.g., NADH, Luciferin, Fluorogenic Probe) | Enables quantitation of reaction velocity. Choice dictates assay format (absorbance, fluorescence, luminescence). |
| 384/96-Well Microplates (Low Binding, Optical Grade) | Platform for high-throughput IC₅₀ determination and substrate-inhibitor matrix experiments. |
| Automated Liquid Handling System | Ensures precision and reproducibility in serial dilutions of inhibitors and substrates, reducing human error. |
| Data Analysis Software (e.g., GraphPad Prism) | Required for nonlinear regression, global curve fitting, and statistical comparison of different inhibition models. |
The Central Role of Substrate/Radioligand Concentration ([S] or [L]) and its Km/Kd
Within the critical research on the Cheng-Prusoff equation for converting IC₅₀ to Kᵢ values, the accurate determination and application of substrate ([S]) or radioligand ([L]) concentration, relative to its Michaelis constant (Kₘ) or dissociation constant (Kd), is paramount. The Cheng-Prusoff derivations (Cheng & Prusoff, 1973) explicitly state that the observed IC₅₀ for a competitive inhibitor is dependent on these parameters. Mischaracterization of [L] or inaccurate Kd measurements are primary sources of error in reported Kᵢ values, impacting drug discovery priorities. These Application Notes detail protocols for defining these core parameters.
Core Quantitative Data & Relationships
Table 1: Cheng-Prusoff Equations for Common Inhibition Mechanisms
| Inhibition Mechanism | Equation for Kᵢ | Critical Concentration Parameter |
|---|---|---|
| Competitive (Enzyme) | ( Ki = \frac{IC{50}}{1 + \frac{[S]}{K_m}} ) | [S], Substrate Concentration |
| Competitive (Receptor) | ( Ki = \frac{IC{50}}{1 + \frac{[L]}{K_d}} ) | [L], Radioligand Concentration |
| Non-Competitive | ( Ki = IC{50} ) | Independent of [S] |
| Uncompetitive (Enzyme) | ( Ki = \frac{IC{50}}{1 + \frac{[S]}{K_m}} ) | [S], Substrate Concentration |
Table 2: Impact of [L]/K_d Ratio on IC₅₀ to Kᵢ Conversion Error
| [L] / K_d | IC₅₀ / Kᵢ (Competitive) | % Error in Kᵢ if [L]/K_d is Underestimated by 50% |
|---|---|---|
| 0.1 | 1.1 | -4.5% |
| 1.0 | 2.0 | -25% |
| 2.0 | 3.0 | -33% |
| 5.0 | 6.0 | -40% |
| 10.0 | 11.0 | -43% |
Experimental Protocols
Protocol 1: Determination of Radioligand K_d via ~12-PointSaturation Binding Assay
Objective: Accurately determine the equilibrium dissociation constant (Kd) and total receptor density (Bmax) for the radioligand used in subsequent inhibition assays. Materials: See "Scientist's Toolkit" below. Procedure:
- Membrane Preparation: Prepare cell membranes expressing the target receptor. Determine total protein concentration (e.g., Bradford assay).
- Dilution Series: Prepare 12 serial dilutions of the radioligand (e.g., [³H]Ligand) in assay buffer, typically spanning from ~0.1 x to 10 x the estimated K_d. Include a high concentration for defining non-specific binding (NSB).
- Binding Reaction: In triplicate, combine in a 96-well plate:
- 50 µL radioligand solution (varying concentration).
- 50 µL membrane suspension (constant protein amount).
- 50 µL assay buffer (for total binding) or 50 µL of a high-concentration unlabeled competitor (e.g., 10 µM, for NSB wells).
- Equilibrium Incubation: Incubate at the appropriate temperature (often 25°C or 4°C) for a duration confirmed to reach equilibrium (≥ 4 x ligand t₁/₂ for association; typically 60-120 min).
- Separation & Quantification: Terminate the reaction by rapid filtration onto GF/B filter plates pre-soaked in 0.3% PEI. Wash filters 3x with ice-cold buffer. Dry filters, add scintillation cocktail, and count on a MicroBeta or similar plate reader.
- Data Analysis: Subtract NSB from total binding at each point to obtain specific binding. Fit specific binding data (Y) vs. radioligand concentration [L] (X) to a one-site specific binding model: ( Y = B{max} * X / (Kd + X) ). The fitted K_d is the critical parameter for Cheng-Prusoff corrections.
Protocol 2: IC₅₀ Determination with Optimized [L]
Objective: Measure the concentration of a test inhibitor that reduces specific radioligand binding by 50%, under a defined [L] condition. Procedure:
- Select [L]: Based on the Kd from Protocol 1, choose a [L] close to its Kd (e.g., [L] = K_d) to balance signal-to-noise with acceptable Cheng-Prusoff shift. Record this value precisely.
- Inhibitor Dilution: Prepare a 10-point, 1:3 serial dilution of the test compound in DMSO, then dilute in assay buffer (final DMSO ≤ 1%).
- Binding Reaction: In triplicate, combine:
- 25 µL inhibitor dilution or buffer/vehicle control.
- 25 µL radioligand solution at 2x the desired final concentration (e.g., 2 * K_d).
- 50 µL membrane suspension.
- Incubation & Quantification: Proceed as in Protocol 1, steps 4-5.
- Data Analysis: Calculate % specific binding relative to vehicle control (100%) and NSB wells (0%). Fit the log(inhibitor) vs. response curve using a 4-parameter logistic model to obtain the IC₅₀ value.
Protocol 3: Kᵢ Calculation via Cheng-Prusoff
Procedure:
- Input Parameters: Obtain the experimental IC₅₀ (from Protocol 2), the precise [L] used, and the radioligand K_d (from Protocol 1).
- Calculation: Apply the competitive Cheng-Prusoff equation: ( Ki = \frac{IC{50}}{1 + \frac{[L]}{K_d}} ).
- Report: Always report the calculated Kᵢ along with the IC₅₀, [L], and K_d used in the calculation.
Visualizations
Title: Workflow from Kd & IC50 to Ki
Title: Competitive Binding Equilibrium & Ki
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions
| Item | Function & Rationale |
|---|---|
| Cell Membranes | Expressing the target receptor (GPCR, kinase, etc.). Source of the binding protein for the assay. |
| Tritiated (³H) or Iodinated (¹²⁵I) Ligand | High-affinity, specific radioligand for the target. Provides the detectable signal for binding measurements. |
| Unlabeled Competitor (e.g., Reference Antagonist) | Used at high concentration to define non-specific binding (NSB) in saturation and inhibition assays. |
| Assay Buffer (with ions, protease inhibitors) | Maintains physiological pH and ionic strength, preserves protein integrity, and minimizes non-specific binding. |
| GF/B Filter Plates & 0.3% PEI Solution | For rapid separation of bound from free radioligand via vacuum filtration. PEI pre-soak reduces ligand adherence to filters. |
| Microplate Scintillation Cocktail | For homogeneous or filter plate-based detection of beta-emitting isotopes (³H, ³⁵S). |
| Liquid Scintillation / MicroBeta Counter | Instrument for quantifying radioactivity (CPM/DPM) from each well to generate binding data. |
| Non-Linear Regression Software (e.g., GraphPad Prism) | Essential for accurately fitting saturation binding (one-site hyperbola) and inhibition data (logistic curve) to derive Kd, Bmax, and IC₅₀. |
Within the broader thesis research on the Cheng-Prusoff equation for converting IC50 to Ki values, a critical and often overlooked step is the accurate determination of the IC50 itself. The Cheng-Prusoff derivation (Ki = IC50 / (1 + [S]/Km)) assumes that the measured IC50 is a true reflection of inhibitor potency under defined, equilibrium conditions. However, IC50 is not an immutable constant; it is a shifting parameter highly dependent on experimental conditions. This application note provides protocols and visual frameworks to systematically quantify and visualize these shifts, ensuring that the input for Ki conversion is robust and contextually understood. Misinterpretation of condition-dependent IC50 shifts is a major source of error in mechanistic enzymology and drug discovery.
Key Experimental Variables Affecting IC50
The following table summarizes primary experimental variables and their documented impact on observed IC50 values for a competitive enzyme inhibitor.
Table 1: Experimental Variables and Their Impact on IC50
| Variable | Typical Range Tested | Direction of IC50 Shift (Competitive Inhibitor) | Mechanistic Reason | Relevance to Cheng-Prusoff |
|---|---|---|---|---|
| Substrate Concentration ([S]) | 0.1x Km to 10x Km | Increases linearly with [S] | Fundamental to competitive inhibition. IC50 = Ki(1+[S]/Km). | Directly accounted for in the equation. |
| Enzyme Concentration ([E]) | 0.1x to 10x standard assay [E] | Increases if [E] > [I]; no shift if [I] >> [E] | Tight-binding conditions violate classical assumptions. | Cheng-Prusoff is invalid under tight-binding conditions. Requires alternate equations (Morrison’s equation). |
| Pre-Incubation Time | 0 to 60 min (or more) | Decreases with longer pre-incubation | Time to reach equilibrium for slow-binding inhibitors. | IC50 at equilibrium is required. Non-equilibrium values underestimate Ki. |
| Assay pH | pH 6.0 to 9.0 | Can increase or decrease | Alters ionization states of inhibitor, substrate, or enzyme active site residues. | Ki should be determined at physiologically/pathologically relevant pH. |
| Cofactor/Ion Concentration | Varying physiological ranges | Variable | May be required for inhibitor binding or compete with inhibitor. | Must be reported and standardized. Can be treated as a competing ligand in extended models. |
| Temperature | 4°C to 37°C | Variable (often decreases with T) | Affects binding kinetics, enzyme conformation, and assay viscosity. | Standardization (e.g., 25°C or 37°C) is critical for reproducibility. |
| ATP Concentration (Kinase Assays) | 1 µM to 1 mM | Increases with [ATP] | ATP is the varied substrate in a bisubstrate reaction. | Requires bisubstrate Cheng-Prusoff type corrections. |
Core Protocol: Systematic IC50 Shift Analysis
This protocol outlines a standardized method to visualize the relationship between a key variable (e.g., substrate concentration) and IC50.
Materials & Reagents
Research Reagent Solutions Toolkit
| Item | Function & Specification |
|---|---|
| Purified Target Enzyme | Recombinant, >90% pure. Aliquoted and stored at -80°C. Concentration accurately determined (A280 or activity assay). |
| Inhibitor Stock Solution | 10 mM in 100% DMSO. Serial dilutions prepared in DMSO to maintain constant [DMSO] (<1% final) across assay. |
| Substrate Stock Solution | Prepared at 10x the highest final concentration needed in assay buffer. Stability under assay conditions verified. |
| Assay Buffer (10X) | Contains all salts, cofactors, and buffering agents at pH-adjusted optimum. Sterile filtered. |
| Detection Reagent | e.g., ATP detection reagent for luciferase-coupled kinase assays, or chromogenic/fluorogenic substrate. Prepared fresh or as per manufacturer. |
| Positive Control Inhibitor | Well-characterized inhibitor with known IC50/Ki under standard conditions. Used for plate and protocol validation. |
| Low-Binding Microplates | 384-well or 96-well plates to minimize compound adsorption. |
| Multichannel Pipettes & Liquid Handler | For accurate, high-throughput reagent dispensing. |
Step-by-Step Methodology
Part A: Multi-Condition Dose-Response Experiment
- Variable Selection: Choose one primary variable to modulate (e.g., [S]). Define 5-6 levels spanning the relevant range (e.g., 0.25xKm, 0.5xKm, 1xKm, 2xKm, 4xKm).
- Plate Layout: Design a plate map where each row contains a full inhibitor dilution series (e.g., 10 concentrations, 1:3 serial dilution). Each column will correspond to a different level of the test variable.
- Reagent Dispensing: a. Add varying concentrations of the test variable (e.g., different [S] in buffer) to the appropriate columns. b. Using a liquid handler, transfer a fixed volume of inhibitor dilution series across the plate rows. c. Initiate all reactions simultaneously by adding a fixed volume of enzyme solution (in the same variable-adjusted buffer) to all wells. For pre-incubation studies, add enzyme before substrate. d. Incubate at constant temperature for the predetermined reaction time. e. Quench the reaction or add detection reagent as required by the assay chemistry.
- Data Acquisition: Read signal (e.g., fluorescence, luminescence) on a plate reader.
Part B: Data Analysis & Visualization
- Normalization: For each test variable column, normalize data: %Inhibition = 100 * (1 – (Signal_inh – Signal_min)/(Signal_max – Signal_min)).
- Curve Fitting: Fit normalized dose-response data for each condition to a 4-parameter logistic (4PL) model: Y = Bottom + (Top-Bottom)/(1+10^((LogIC50-X)*HillSlope)).
- IC50 Extraction: Record the fitted IC50 and its confidence interval for each condition.
- Shift Visualization: Create two key plots: a. Overlaid Dose-Response Curves: Visually show the right- or left-shift. b. IC50 vs. Variable Plot: Plot extracted IC50 values (log scale) against the variable (e.g., [S]/Km). For a competitive inhibitor, fit a line to IC50 = Ki(1 + [S]/Km) to estimate Ki.
Visualization of Concepts and Workflows
Application Note: Case Study – A Kinase Inhibitor
Objective: To determine the true Ki of a novel ATP-competitive kinase inhibitor and validate the Cheng-Prusoff relationship.
Protocol Execution:
- The assay was run at five [ATP] levels: 5 µM, 15 µM, 45 µM (≈Km), 135 µM, and 405 µM.
- A 10-point inhibitor dilution series was tested in quadruplicate at each [ATP].
- Data were analyzed as per Section 3.2.
Results & Visualization: Table 2: Observed IC50 Shifts with Varying [ATP]
| [ATP] (µM) | [ATP]/Km | Observed IC50 (nM) | 95% CI (nM) | Calculated Ki (nM)* |
|---|---|---|---|---|
| 5 | 0.11 | 12.1 | 10.5 – 13.9 | 10.9 |
| 15 | 0.33 | 18.5 | 16.2 – 21.1 | 12.4 |
| 45 | 1.00 | 38.7 | 34.1 – 43.9 | 19.4 |
| 135 | 3.00 | 112.3 | 98.8 – 127.7 | 28.1 |
| 405 | 9.00 | 401.5 | 352.0 – 458.0 | 40.2 |
*Using Cheng-Prusoff: Ki = IC50 / (1 + [ATP]/Km), assuming Km(ATP)=45 µM.
Interpretation: The IC50 increased linearly with [ATP], confirming ATP-competitive mechanism. However, the calculated Ki was not constant but increased at high [ATP]/Km ratios, suggesting a potential deviation from the simple competitive model (e.g., inhibitor binding affects affinity for ATP) or assay limitations at high [ATP]. This visualization flags the need for a more sophisticated binding model beyond the basic Cheng-Prusoff approximation for accurate Ki determination across all relevant physiological ATP concentrations.
Systematic visualization of IC50 shifts is not merely a quality control step; it is a fundamental mechanistic investigation. For research centered on the Cheng-Prusoff equation, these protocols enable researchers to:
- Identify the appropriate assay conditions for valid IC50 measurement.
- Provide visual proof of the inhibition modality (competitive, non-competitive).
- Flag conditions where Cheng-Prusoff approximations break down (e.g., tight-binding, allosteric inhibition).
- Generate the robust, condition-aware IC50 data required for accurate Ki conversion, thereby strengthening the entire foundational thesis on enzyme inhibitor characterization.
Step-by-Step Guide: Applying the Cheng-Prusoff Equation in Your Research
Accurate determination of biochemical constants is the cornerstone of rigorous enzyme and receptor pharmacology. This protocol, framed within the broader thesis on Cheng-Prusoff equation research, provides detailed methodologies for obtaining the precise experimental values (IC₅₀, Kₘ, K𝒹, and [S]/[L]) required for the valid conversion of IC₅₀ to the inhibition constant (Kᵢ). The Cheng-Prusoff relationship (Kᵢ = IC₅₀ / (1 + [S]/Kₘ) for competitive inhibitors) is wholly dependent on the accuracy of these input parameters. This document serves as a critical guide for researchers and drug development professionals to establish robust foundational data.
Part 1: Experimental Protocols for Determining Key Parameters
Protocol 1.1: Determination of Enzyme Michaelis Constant (Kₘ) and Vmax
Objective: To characterize enzyme kinetics under initial velocity conditions and determine Kₘ, the substrate concentration at half-maximal velocity.
- Reagent Setup: Prepare a master mix containing assay buffer, cofactors, and a fixed, limiting concentration of enzyme. Aliquot this into a microplate.
- Substrate Titration: Add substrate solution across a series of wells to create a concentration range typically spanning 0.2 to 5 times the estimated Kₘ (e.g., 8 concentrations in duplicate).
- Reaction Initiation & Monitoring: Initiate the reaction uniformly. Monitor product formation continuously (kinetic read) or stop the reaction at a fixed, early time point (endpoint read) where less than 10% of substrate is consumed.
- Data Analysis: Plot initial velocity (v) against substrate concentration ([S]). Fit data to the Michaelis-Menten equation: v = (Vmax * [S]) / (Kₘ + [S]) using nonlinear regression. Do not rely on linearized plots (e.g., Lineweaver-Burk) for final parameter estimation.
Protocol 1.2: Determination of Ligand-Receptor Dissociation Constant (K𝒹) via Saturation Binding
Objective: To quantify the affinity of a labeled ligand for its receptor.
- Membrane/Receptor Preparation: Isolate cell membranes expressing the target receptor or use purified receptor protein.
- Binding Reaction: Incubate a fixed concentration of receptor with increasing concentrations of radiolabeled or fluorescent ligand ([L]) in binding buffer. Include parallel wells with a large excess of unlabeled competitor to define non-specific binding (NSB).
- Separation & Quantification: Separate bound from free ligand via filtration, centrifugation, or other appropriate methods. Quantify bound labeled ligand.
- Data Analysis: Calculate specific binding = Total Binding - NSB. Plot specific bound ligand ([Bound]) vs. free ligand concentration ([Free]). Fit data to a one-site specific binding model: [Bound] = (Bmax * [Free]) / (K𝒹 + [Free]), where Bmax is total receptor density.
Protocol 1.3: Determination of Inhibitor IC₅₀
Objective: To measure the concentration of inhibitor that reduces enzyme activity or ligand binding by 50% under a defined set of conditions.
- Condition Setup: Establish optimal assay conditions from Protocols 1.1 or 1.2. Use a single, fixed concentration of substrate ([S]) near its Kₘ value or labeled ligand ([L]) near its K𝒹 value.
- Inhibitor Titration: Serially dilute the test inhibitor and add to the reaction. Include controls for 100% activity (no inhibitor) and 0% activity (background/blank).
- Assay Execution: Perform the reaction as per the established kinetic or binding protocol.
- Data Analysis: Normalize response data to % activity or % binding. Plot % activity vs. log[Inhibitor]. Fit data to a 4-parameter logistic (sigmoidal) model: Y = Bottom + (Top-Bottom) / (1 + 10^((LogIC₅₀ - X)*HillSlope)) to derive the IC₅₀.
Protocol 1.4: Accurate Measurement of Assay [S] and [L]
Objective: To verify the exact, free concentration of substrate or ligand present during the IC₅₀ experiment.
- For [S]: The nominal concentration from preparation is often sufficient for soluble enzymes. For cell-based assays, verify substrate uptake/permeability. Critical control: Ensure initial velocity conditions where substrate depletion is minimal (<10%).
- For [L] (Binding Assays): The free concentration ([Free]) is nominal total ligand minus bound ligand. In IC₅₀ experiments where [L] ≈ K𝒹, significant receptor depletion can occur, altering free concentration. Rule: If receptor concentration ([R]) > 0.1 * K𝒹, use the quadratic solution to calculate [Free], or design the assay to keep [R] << K𝒹 to use nominal [L].
Part 2: Data Presentation
| Parameter | Definition | Typical Assay | Critical Controls for Accuracy | Impact on Cheng-Prusoff |
|---|---|---|---|---|
| IC₅₀ | Half-maximal inhibitory concentration | Inhibitor dose-response under fixed conditions | Solvent/DMSO controls, signal window (Z'), compound interference (fluorescence, quenching) | Direct input variable. Must be from a robust, high-quality curve. |
| Kₘ | Michaelis constant; [S] at ½ Vmax | Substrate velocity kinetics | Substrate depletion (<10%), linear initial rates, enzyme stability | Used in denominator. Error propagates significantly into Kᵢ. |
| K𝒹 | Dissociation constant; [L] at ½ Bmax | Saturation binding isotherm | Non-specific binding definition, ligand/receptor depletion, filtration efficiency | Used to calculate [L]/K𝒹 for binding assays (Cheng-Prusoff: Kᵢ = IC₅₀ / (1 + [L]/K𝒹)). |
| [S] | Free substrate concentration in IC₅₀ assay | Calculated from preparation and consumption | Verify via HPLC/MS or enzyme-coupled assays if unstable. Ensure constant [S] during measurement. | Must be the true free concentration during the IC₅₀ experiment. |
| [L] | Free labeled ligand concentration in IC₅₀ assay | Calculated (Nominal - Bound) | Account for receptor depletion if [R] is high relative to K𝒹. | Must be the true free concentration during the IC₅₀ experiment. |
Table 2: Common Pitfalls and Validation Steps
| Parameter | Common Pitfall | Consequence | Validation Step |
|---|---|---|---|
| IC₅₀ | Inadequate curve span (top/bottom plateaus not defined) | Inaccurate IC₅₀ estimate | Test inhibitor from 0% to 100% effect; use ≥10 concentrations spanning expected IC₅₀. |
| Kₘ | Assaying under non-initial velocity conditions | Underestimation of Kₘ and Vmax | Measure progress curves; ensure linear product formation over assay time. |
| K𝒹 | Inaccurate determination of non-specific binding | Systematic error in K𝒹 and Bmax | Use a high-affinity unlabeled competitor at 100-1000x its Kᵢ to define NSB. |
| [S]/[L] | Ignoring depletion (ligand or substrate) | [S]free or [L]free ≠ [S]total or [L]total | Calculate % depletion. If >10%, use quadratic binding equation or lower receptor/enzyme concentration. |
Part 3: The Scientist's Toolkit
Research Reagent Solutions & Essential Materials
| Item | Function in Determining Prerequisites |
|---|---|
| High-Purity Enzyme/Receptor | Target protein with confirmed activity and minimal contamination to ensure specific signal. |
| Characterized Substrate/Ligand | Molecule with known purity and stability. For ligands, specific activity of labeled tracer is critical. |
| Validated Inhibitor Compounds | Stocks prepared in DMSO or buffer, with concentration verified (e.g., by UV absorbance). |
| Homogeneous Assay Platform | (e.g., Fluorescence Polarization, TR-FRET, AlphaScreen). Enables rapid, separation-free measurement of binding/activity for high-quality kinetics. |
| Radioisotopic Tracers (e.g., ³H, ¹²⁵I) | Traditional high-sensitivity ligands for saturation and competition binding to determine K𝒹 and Bmax. |
| Microplate Reader with Kinetics | Instrument capable of precise spectrophotometric or fluorometric reads over time for kinetic assays. |
| Liquid Handling Robot | For accurate and reproducible serial dilutions of substrates/inhibitors, minimizing pipetting error. |
| Nonlinear Regression Software | (e.g., GraphPad Prism, SigmaPlot). Essential for robust fitting of data to Michaelis-Menten, binding isotherm, and dose-response models. |
Part 4: Visualizations
Diagram 1: Workflow for Accurate Ki Determination
Diagram 2: Cheng-Prusoff Derivation & Assumptions
Within the broader research on the Cheng-Prusoff equation for converting IC₅₀ to Kᵢ values, a critical preliminary step is the correct identification of the inhibition model governing the interaction under study. The Cheng-Prusoff derivations assume specific conditions (e.g., rapid equilibrium, single inhibitor binding site, no substrate depletion). Applying an incorrect equation form based on a mismatched model introduces significant error into Kᵢ determination, compromising drug development decisions. These application notes provide a structured approach to match experimental assay data to the correct inhibition model and its corresponding equation form.
Theoretical Framework: Inhibition Models and Equations
Inhibitor-enzyme interactions are classified based on the site of inhibition relative to the substrate and the binding kinetics. The primary models are Competitive, Non-Competitive, Uncompetitive, and Mixed Inhibition. Each has a distinct effect on the Michaelis-Menten parameters (Kₘ and Vₘₐₓ) and requires a unique form of the Cheng-Prusoff correction.
Table 1: Inhibition Models, Characteristics, and Corrected Cheng-Prusoff Equations
| Inhibition Model | Binding Site Relative to Substrate | Effect on Apparent Kₘ (Kₘₐₚₚ) | Effect on Apparent Vₘₐₓ (Vₘₐₓₐₚₚ) | Conversion Equation (Kᵢ from IC₅₀) |
|---|---|---|---|---|
| Competitive | Same (active site) | Increases: Kₘₐₚₚ = Kₘ(1 + [I]/Kᵢ) | Unchanged: Vₘₐₓₐₚₚ = Vₘₐₓ | Kᵢ = IC₅₀ / (1 + [S]/Kₘ) |
| Non-Competitive | Different (allosteric) | Unchanged: Kₘₐₚₚ = Kₘ | Decreases: Vₘₐₓₐₚₚ = Vₘₐₓ/(1 + [I]/Kᵢ) | Kᵢ = IC₅₀ / (1 + [S]/Kₘ) Note: True only for α=∞ |
| Uncompetitive | Enzyme-Substrate Complex only | Decreases: Kₘₐₚₚ = Kₘ/(1 + [I]/αKᵢ) | Decreases: Vₘₐₓₐₚₚ = Vₘₐₓ/(1 + [I]/Kᵢ) | Kᵢ = IC₅₀ / (1 + ([S]/Kₘ)) |
| Mixed | Different, affects substrate binding | Increases or Decreases | Decreases | Kᵢ = IC₅₀ / (1 + ([S]/αKₘ)) *α is the factor by which inhibitor binding alters substrate affinity. |
Note: [S] = substrate concentration used in the IC₅₀ assay; Kₘ = Michaelis constant determined for the enzyme under identical assay conditions.
Experimental Protocol: Determining the Inhibition Model
Objective: To conduct a series of initial velocity experiments that characterize the mode of inhibition, enabling the selection of the correct Kᵢ conversion equation.
Materials & Reagents: See "The Scientist's Toolkit" below. Workflow Overview:
- Determine Kₘ and Vₘₐₓ for the uninhibited reaction.
- Measure initial reaction velocities at varying substrate concentrations in the presence of several fixed inhibitor concentrations.
- Plot and analyze data using Lineweaver-Burk (double reciprocal) and Dixon plots.
- Identify the pattern to classify the inhibition model.
Detailed Protocol:
- Kₘ and Vₘₐₓ Determination:
- Prepare a dilution series of the substrate (typically 6-8 concentrations spanning 0.2Kₘ to 5Kₘ).
- Initiate reactions by adding a fixed concentration of enzyme. Measure initial velocity (v₀) for each [S] via absorbance, fluorescence, or radioactivity.
- Fit the data (v₀ vs. [S]) to the Michaelis-Menten equation using nonlinear regression software to obtain Kₘ and Vₘₐₓ.
Inhibition Matrix Experiment:
- Prepare reactions with the same substrate concentration series as in Step 1.
- For each substrate concentration, run parallel reactions with at least four different inhibitor concentrations (e.g., 0, 0.5×IC₅₀, 1×IC₅₀, 2×IC₅₀) and a negative control (no enzyme).
- Perform all reactions in triplicate.
Data Analysis & Model Identification:
- Lineweaver-Burk Plot (1/v vs. 1/[S]): Plot data for each inhibitor concentration.
- Competitive: Lines intersect on the y-axis (1/Vₘₐₓ unchanged).
- Non-Competitive: Lines intersect on the x-axis (1/Kₘ unchanged).
- Uncompetitive: Parallel lines.
- Mixed: Lines intersect in the left quadrant.
- Dixon Plot (1/v vs. [I]): For a single, fixed [S], plot data from multiple inhibitor concentrations. The intersection point provides an estimate of -Kᵢ.
- Perform global nonlinear regression fitting of the full dataset (v₀ vs. [S], [I]) to each inhibition model equation. Use statistical criteria (e.g., AICc, R², residual analysis) to select the best-fit model.
- Lineweaver-Burk Plot (1/v vs. 1/[S]): Plot data for each inhibitor concentration.
Pathway and Workflow Visualizations
Title: Workflow for Matching Assay Data to Inhibition Model
Title: Key Enzyme-Inhibitor-Substrate Binding Schemes
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for Inhibition Modeling
| Item/Reagent | Function & Rationale |
|---|---|
| Recombinant Target Enzyme | High-purity, well-characterized enzyme is essential for reproducible kinetic measurements and accurate Kₘ determination. |
| Natural Substrate or Fluorogenic/Chromogenic Substrate Analog | To monitor reaction progress. The analog must have validated kinetic parameters (kcat, Kₘ) similar to the natural substrate. |
| Inhibitor Compound(s) | Compounds of interest dissolved in appropriate solvent (e.g., DMSO) at high concentration, with solvent concentration normalized (<1% v/v) across all reactions. |
| Cofactor/Coenzyme Solutions | Required for the enzymatic reaction (e.g., Mg²⁺ for kinases, NADPH for reductases). Must be included at saturating concentrations. |
| Activity Assay Buffer | Optimized for pH, ionic strength, and stability. Often includes BSA or reducing agents to maintain enzyme activity. |
| Stop Solution or Detection Reagent | To quench the reaction at precise timepoints or to generate a detectable signal (e.g., malachite green for phosphate, coupled enzyme systems). |
| Microplate Reader (Absorbance/Fluorescence/Luminescence) | For high-throughput acquisition of initial velocity data from multi-well plates. |
| Nonlinear Regression Analysis Software | Essential for robust fitting of data to Michaelis-Menten and inhibition models (e.g., GraphPad Prism, SigmaPlot, R). |
Accurate conversion of IC₅₀ to the absolute affinity constant Kᵢ is not a mere algebraic exercise but a model-dependent process. Systematic experimental determination of the inhibition mechanism prior to applying the Cheng-Prusoff correction is non-negotiable for reliable Kᵢ reporting in drug discovery. The protocols and framework provided herein ensure researchers can match their assay data to the correct inhibition model, thereby upholding the integrity of downstream pharmacological analyses and compound optimization efforts.
This protocol provides a detailed, practical walkthrough for conducting a competitive enzyme inhibition assay and applying the Cheng-Prusoff equation to derive the inhibition constant (Kᵢ). It is situated within a broader thesis research framework examining the proper application and limitations of the Cheng-Prusoff equation for converting half-maximal inhibitory concentration (IC₅₀) values to Kᵢ values. Accurate determination of Kᵢ is fundamental in enzymology and drug discovery for comparing inhibitor potency irrespective of assay conditions.
Key Research Reagent Solutions
| Reagent/Material | Function in Experiment |
|---|---|
| Recombinant Target Enzyme | The protein whose activity is being modulated; purity and stability are critical. |
| Fluorogenic/Chromogenic Substrate | Compound converted by the enzyme to a measurable signal (absorbance/fluorescence). |
| Test Inhibitor Compound | The molecule whose inhibitory potency is being quantified. |
| Assay Buffer (with Mg²⁺/ATP if kinase) | Maintains optimal pH, ionic strength, and cofactor conditions for enzyme activity. |
| Positive Control Inhibitor (Known Kᵢ) | Validates assay performance and calculation methodology. |
| Microplate Reader-Compatible Plates | Vessel for conducting high-throughput reaction measurements. |
| DMSO (High Purity) | Universal solvent for stock solutions of lipophilic inhibitors; keep concentration constant (<1%). |
Experimental Protocol: Competitive Inhibition Assay
Preliminary Setup
- Prepare Inhibitor Dilutions: Create a serial dilution (e.g., 1:3) of the test compound in DMSO, typically spanning 10 concentrations. Use the same DMSO concentration in all wells, including controls.
- Prepare Substrate Solution: Make a stock solution of the substrate in assay buffer. The final concentration in the assay should be at or below its Michaelis constant (Kₘ) for reliable Cheng-Prusoff application.
- Prepare Enzyme Solution: Dilute the recombinant enzyme in cold assay buffer to the working concentration.
Reaction Setup (96-Well Plate Format)
- Dispense Inhibitor: Add 2 µL of each inhibitor dilution (or DMSO control) to the appropriate wells in triplicate.
- Add Enzyme: Pipette 38 µL of the enzyme solution into all wells containing inhibitor. Incubate for 15 minutes at assay temperature to pre-bind enzyme-inhibitor.
- Initiate Reaction: Start the reaction by adding 40 µL of the substrate solution to all wells using a multichannel pipette. Final reaction volume is 80 µL.
- Kinetic Measurement: Immediately transfer the plate to a pre-warmed microplate reader. Record the increase in fluorescence/absorbance every 30 seconds for 15-30 minutes.
Data Collection & IC₅₀ Determination
- Calculate the initial reaction velocity (V₀) for each well from the linear slope of the signal vs. time plot.
- Normalize V₀ as a percentage of the average DMSO-only control (0% inhibition) and blank-subtracted baseline (100% inhibition).
- Plot inhibitor concentration [I] (log scale) vs. % inhibition or % activity.
- Fit the data to a four-parameter logistic (4PL) model (variable slope) to determine the IC₅₀ value—the concentration yielding 50% inhibition.
Data and Calculations: Applying the Cheng-Prusoff Equation
Table 1: Experimental Parameters and Derived IC₅₀
| Parameter | Symbol | Value | Note |
|---|---|---|---|
| Substrate Concentration | [S] | 10 µM | Must be known and fixed. |
| Michaelis Constant | Kₘ | 15 µM | Determined in separate saturation kinetics experiment. |
| Measured IC₅₀ | IC₅₀ | 2.5 µM | From dose-response curve fitting. |
| Enzyme Inhibition Mode | — | Competitive | Must be confirmed mechanistically. |
Table 2: Cheng-Prusoff Calculation for Competitive Inhibition
| Step | Formula | Calculation | Result (Kᵢ) |
|---|---|---|---|
| Cheng-Prusoff Equation | Kᵢ = IC₅₀ / (1 + ([S]/Kₘ)) | ||
| 1. Calculate [S]/Kₘ ratio | [S] / Kₘ | 10 µM / 15 µM | 0.667 |
| 2. Calculate 1 + ([S]/Kₘ) | 1 + ([S]/Kₘ) | 1 + 0.667 | 1.667 |
| 3. Compute Kᵢ | IC₅₀ / 1.667 | 2.5 µM / 1.667 | 1.5 µM |
Interpretation: The apparent potency (IC₅₀) of 2.5 µM, measured at [S]=10 µM, corresponds to a true dissociation constant (Kᵢ) of 1.5 µM for the enzyme-inhibitor complex.
Title: Workflow for Converting IC50 to Ki
Title: Competitive Inhibition Mechanism & Constants
Application Notes: Software Evolution in Ki Determination
The accurate conversion of IC₅₀ values to equilibrium inhibition constants (Kᵢ) via the Cheng-Prusoff equation is foundational to enzymology and drug discovery. This process has evolved from manual, error-prone calculations to integrated, automated platforms, enhancing reproducibility and throughput.
Quantitative Comparison of Analysis Platforms
The table below compares key software tools used for Kᵢ analysis, highlighting their evolution from manual systems to automation.
Table 1: Comparison of Software Tools for IC₅₀ to Kᵢ Analysis
| Tool Category | Example Tools | Primary Function | Automation Level | Key Advantage | Typical Data Output |
|---|---|---|---|---|---|
| Manual Spreadsheets | Microsoft Excel, Google Sheets | Manual data entry & formula application | Low | High flexibility, low cost | IC₅₀, Kᵢ (single values) |
| Curve-Fitting Software | GraphPad Prism, SigmaPlot | Nonlinear regression for dose-response | Medium | Robust statistical validation | IC₅₀ ± SEM, curve plots |
| Automated Analysis Suites | Genedata Screener, Dotmatics | High-throughput screening data management | High | Integrated workflow, audit trail | IC₅₀, Kᵢ, Z'-factor, full reports |
| Specialized Kᵢ Platforms | assayQuant, KinTek KINSIM | Mechanistic modeling & precise Kᵢ calculation | High | Accounts for complex kinetics | Kᵢ ± confidence interval, model parameters |
Critical Considerations for Platform Selection
- Equation Form: Ensure the software implements the correct Cheng-Prusoff derivation (e.g., for competitive inhibition: Kᵢ = IC₅₀ / (1 + [S]/Kₘ)).
- Error Propagation: Advanced tools propagate error from IC₅₀ and Kₘ measurements to report confidence intervals for Kᵢ.
- Substrate Concentration ([S]) & Kₘ Accuracy: The quality of Kᵢ data is directly dependent on the accurate determination of these parameters.
Experimental Protocols
Protocol: Determination of IC₅₀ Using a Fluorescence-Based Assay
Aim: To generate a dose-response curve for a novel inhibitor against target enzyme 'X' and determine the IC₅₀ value.
Research Reagent Solutions & Essential Materials: Table 2: Key Reagents for Enzymatic IC₅₀ Assay
| Item | Function | Example/Notes |
|---|---|---|
| Recombinant Enzyme X | The pharmacological target. | Purified, >95% purity; store at -80°C. |
| Fluorogenic Substrate | Enzyme activity reporter. | Must be selective for Enzyme X. Km pre-determined. |
| Test Inhibitor | Compound for characterization. | 10 mM stock in DMSO; serial dilute in assay buffer. |
| Assay Buffer | Maintains optimal enzyme activity. | Typically 50 mM Tris-HCl, pH 7.5, 10 mM MgCl₂. |
| Positive Control Inhibitor | Validates assay performance. | Known potent inhibitor of Enzyme X. |
| 384-Well Microplate | Reaction vessel for HTS format. | Black, low-volume, flat-bottom plates. |
| Plate Reader | Detects fluorescence signal. | Equipped with appropriate excitation/emission filters. |
Methodology:
- Inhibitor Dilution: Prepare a 3-fold serial dilution of the test inhibitor in assay buffer across 10 concentrations in a separate polypropylene plate. Include a DMSO-only control (0% inhibition) and a well with a high concentration of control inhibitor (100% inhibition).
- Reaction Setup: Using an automated liquid handler, transfer 5 µL of each inhibitor dilution or control into a 384-well assay plate, in triplicate.
- Enzyme Addition: Add 20 µL of Enzyme X (diluted in assay buffer to a pre-optimized concentration) to all wells. Centrifuge briefly at 1000 × g to collect contents.
- Pre-incubation: Incubate plate for 15 minutes at 25°C to allow inhibitor-enzyme equilibrium.
- Reaction Initiation: Initiate the reaction by adding 25 µL of substrate solution (at concentration [S], typically near Kₘ). Final assay volume is 50 µL.
- Kinetic Measurement: Immediately place plate in a plate reader and measure fluorescence (Ex/Em as per substrate) every minute for 30 minutes at 25°C.
- Data Analysis: Calculate initial reaction velocities (V) from the linear phase of progress curves. Normalize V as a percentage of the DMSO control (100% activity) and positive control (0% activity). Fit normalized data to a log(inhibitor) vs. response (variable slope) model: Y = Bottom + (Top-Bottom)/(1+10^((LogIC₅₀-X)HillSlope))* to determine IC₅₀.
Protocol: Conversion of IC₅₀ to Kᵢ Using an Automated Analysis Platform
Aim: To apply the Cheng-Prusoff equation within an automated workflow to calculate Kᵢ, incorporating known experimental parameters and error.
Methodology:
- Data Import: Import the dose-response data (inhibitor concentration, normalized response) and the calculated IC₅₀ value with its standard error (from Protocol 2.1) into the analysis platform (e.g., Genedata Screener).
- Parameter Definition: In the software's analysis template, define the relevant parameters:
- Substrate concentration used in the assay, [S].
- Michaelis constant for the substrate, Kₘ (with its standard error), determined in a separate saturating kinetics experiment.
- Inhibition Model: Select "Competitive" inhibition (the most common assumption for Cheng-Prusoff application).
- Equation Application: Configure the software to apply the competitive Cheng-Prusoff equation: Kᵢ = IC₅₀ / (1 + [S]/Kₘ). Ensure the algorithm is set to propagate errors from both IC₅₀ and Kₘ to calculate a confidence interval (e.g., 95% CI) for the final Kᵢ.
- Batch Processing: Apply this configured template to all inhibitors tested in the same assay batch for consistent, high-throughput Kᵢ determination.
- Report Generation: Export a data table containing: Compound ID, IC₅₀, IC₅₀ SE, [S], Kₘ, Kₘ SE, calculated Kᵢ, and Kᵢ 95% CI.
Visualizations
Title: IC50 to Ki Analysis Workflow
Title: Software Tool Evolution in Ki Analysis
This document establishes best practices for reporting inhibition constant (Ki) values derived from the Cheng-Prusoff equation within competitive binding or functional assays. The broader thesis context posits that while the Cheng-Prusoff equation (Ki = IC50 / (1 + [L]/Kd or 1 + [S]/Km)) remains a foundational tool for converting half-maximal inhibitory concentration (IC50) to Ki, the accurate communication of its output requires rigorous statistical treatment. The Ki value is an estimate, and its reliability must be conveyed through the explicit reporting of confidence intervals (CIs), propagating error from all experimental inputs.
Core Principles for Ki & CI Reporting
Mandatory Parameters to Report:
- Calculated Ki Value: The point estimate.
- Confidence Interval (e.g., 95% CI): The interval estimate.
- Assay Type: Competitive binding vs. functional (enzyme activity, cell signaling).
- Cheng-Prusoff Correction Applied: Specify the exact form (e.g., for competitive binding: Ki = IC50/(1+[L]/Kd)).
- Key Input Values with their CIs/Errors: [L] (radioligand concentration), Kd (ligand-receptor dissociation constant), [S] (substrate concentration), Km (Michaelis constant), and the IC50 itself.
- Statistical Method for CI Propagation: e.g., Error propagation (analytic), Monte Carlo simulation, or bootstrap method.
- Number of Independent Experiments (N): Not replicates.
Table 1: Example Reporting Format for a Competitive Binding Assay
| Parameter | Value | 95% CI / Error | Units | Notes |
|---|---|---|---|---|
| IC₅₀ (Compound X) | 10.0 | [8.2, 12.5] | nM | From 4 independent competition curves. |
| Radioligand [L] | 2.0 | ± 0.1 | nM | Directly measured. |
| Radioligand Kd | 1.5 | [1.3, 1.8] | nM | From saturation binding (3 exp). |
| Cheng-Prusoff Factor | 2.33 | — | — | Calculated as (1 + [L]/Kd). |
| Calculated Ki | 4.29 | [3.2, 5.9] | nM | Via Monte Carlo error propagation (10,000 iterations). |
Table 2: Comparison of CI Propagation Methods
| Method | Principle | When to Use | Key Output |
|---|---|---|---|
| Analytic Error Propagation | Uses derivatives (Gauss's formula) to combine SEs of inputs. | Input errors are small, symmetric, and normally distributed. | Ki SE, used to calculate CI. |
| Monte Carlo Simulation | Randomly samples input parameters from their distributions, recalculates Ki many times. | Input errors are non-normal or large; most robust general method. | Empirical distribution of Ki, direct CI percentiles. |
| Bootstrap | Resamples original dose-response data to generate new IC50 distributions. | To incorporate full uncertainty from curve-fitting of primary data. | CI derived from resampled Ki values. |
Experimental Protocols
Protocol: Determining IC50 with CI for Cheng-Prusoff Input
Objective: Generate a reliable IC50 estimate with confidence interval from a competitive binding or functional dose-response experiment.
Procedure:
- Assay Execution: Perform the inhibition assay (e.g., radioligand binding, enzyme activity) across a minimum of 10 inhibitor concentrations, spaced logarithmically (e.g., half-log increments). Perform each condition in replicate (technical) and repeat in N ≥ 3 independent experiments.
- Data Normalization: For each experiment, normalize response data to percentage of control activity (e.g., 0% = non-specific binding; 100% = total binding in absence of inhibitor).
- Non-Linear Regression: Fit normalized, combined data from a single experiment to a four-parameter logistic (4PL) model:
Response = Bottom + (Top - Bottom) / (1 + 10^((LogIC50 - Log[Inhibitor]) * HillSlope)) - IC50 Extraction: Extract the LogIC50 and its standard error (SE) from the curve fit for each independent experiment.
- Global Fitting (Recommended): Fit data from all N experiments simultaneously using a mixed-effects model or global fitting with shared IC50, sharing the Hill Slope and top/bottom plateaus where appropriate. This yields a more robust global IC50 estimate and CI.
- Report: The global IC50 (geometric mean if combining separate fits) and its 95% CI. Do not use the SE from a single experiment's curve fit.
Protocol: Monte Carlo Simulation for Ki CI Propagation
Objective: Propagate uncertainty from IC50, [L], and Kd to obtain a confidence interval for the calculated Ki.
Procedure:
- Define Input Distributions:
- IC50: Assume log-normal distribution. Use the natural log of the IC50 estimate (ln(IC50)) and its SE from the curve fit.
- Kd and [L]: Assume normal distribution. Use the estimate and its SE (or half the CI width / 1.96).
- Simulation Loop (Iterate 10,000 times):
a. Randomly sample a value for
IC50_simfrom its log-normal distribution. b. Randomly sample values forKd_simandL_simfrom their normal distributions. c. CalculateKi_sim = IC50_sim / (1 + (L_sim / Kd_sim)). d. StoreKi_sim. - Analyze Output: Sort the 10,000
Ki_simvalues. The 2.5th and 97.5th percentiles of this distribution constitute the 95% CI for the Ki. - Report: Present the median of the
Ki_simdistribution as the point estimate and the [2.5%, 97.5%] percentiles as the 95% CI.
Visualizations
Workflow for Ki and Confidence Interval Determination
Competitive Binding Basis for Cheng-Prusoff
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Ki Determination Studies
| Item / Reagent | Function in Ki Determination | Critical Consideration for CI Reporting |
|---|---|---|
| Tritiated or Fluorescent Ligand | The probe ([L]) whose displacement is measured. | Precise concentration determination (via spectrometry) is vital. Error in [L] directly propagates to Ki error. |
| Reference Kd Ligand | Used in separate saturation experiments to determine the Kd of the probe. | High purity required. The CI of the Kd is a mandatory input for error propagation. |
| Cell Membrane Prep or Recombinant Enzyme | Source of the target (receptor or enzyme). | Batch-to-batch variability impacts absolute IC50. Use same preparation for full inhibitor series. |
| Microplate Scintillation/ Fluorescence Reader | Quantifies bound ligand or enzymatic product. | Instrument linearity and dynamic range must be validated to ensure accurate % inhibition calculation. |
| Non-Linear Regression Software (e.g., GraphPad Prism, R) | Fits dose-response data to obtain IC50 & SE/CI. | Must provide standard error or CI of the fitted IC50, not just the point estimate. |
| Statistical Computing Environment (e.g., R, Python with NumPy) | Executes Monte Carlo simulations for error propagation. | Enables robust CI calculation for Ki when input parameter distributions are defined. |
The Cheng-Prusoff equation (Ki = IC50 / (1 + [S]/Km)) is a cornerstone for converting apparent inhibition potency (IC50) to a true binding constant (Ki). This conversion is critical across the drug discovery pipeline, from initial High-Throughput Screening (HTS) to detailed mechanistic studies. Accurate Ki determination validates HTS hits, informs structure-activity relationships (SAR), and enables precise comparison of inhibitor affinity under varying assay conditions, which is fundamental to the thesis on the equation's modern applications and limitations.
Application Notes & Protocols
Application Note: Primary HTS for Kinase Inhibitors
Objective: Identify initial hit compounds from a large library (>100,000 compounds) targeting a tyrosine kinase. Thesis Context: Raw IC50 values from HTS are preliminary. The Cheng-Prusoff correction is applied post-HTS to account for the fixed, often non-saturating, ATP concentration ([S]) used in the screen, converting IC50 to Ki for accurate initial affinity ranking.
Key Quantitative Data (Representative): Table 1: HTS Assay Parameters & Output
| Parameter | Value | Description |
|---|---|---|
| Assay Type | Luminescent (ATP depletion) | Measures kinase activity. |
| Library Size | 150,000 compounds | Diversity-focused chemical library. |
| [ATP] ([S]) | 10 µM | Fixed substrate concentration. |
| Km (ATP) | 15 µM | Experimentally determined for target kinase. |
| Hit Threshold | >70% Inhibition | Initial activity cutoff. |
| Primary Hit Rate | 0.5% (~750 compounds) | Compounds passing threshold. |
| Avg. IC50 of Hits | 0.1 - 10 µM | Range of apparent potency. |
Research Reagent Solutions Toolkit: Table 2: Essential Reagents for Kinase HTS
| Reagent/Kit | Vendor Example | Function |
|---|---|---|
| Recombinant Kinase Protein | Carna Biosciences, SignalChem | Purified target enzyme. |
| ADP-Glo Kinase Assay | Promega | Luminescent, homogenous assay for ADP detection. |
| Substrate Peptide | Eurofins | Kinase-specific peptide (e.g., Poly-Glu-Tyr). |
| Compound Library | ChemBridge, Enamine | Diverse small molecules for screening. |
| 1536-Well Plates | Corning | Microplate format for ultra-HTS. |
| Liquid Handling Robot | Beckman Coulter (Biomek) | Automated compound/reagent dispensing. |
Protocol 2.1.1: HTS Luminescent Kinase Assay
- Plate Preparation: Dispense 2 µL of compound (in DMSO) into 1536-well assay plates using an acoustic dispenser. Include controls (100% activity, 0% activity with staurosporine).
- Enzyme/Substrate Mix: Prepare a solution containing kinase (2 nM final) and peptide substrate (0.2 µg/µL final) in assay buffer.
- Reaction Initiation: Add 3 µL of Enzyme/Substrate mix to plates, pre-incubate for 10 min at 25°C. Initiate reaction by adding 2 µL of ATP solution (10 µM final concentration in well).
- Incubation: Incubate plates for 60 min at 25°C.
- Detection: Add 5 µL of ADP-Glo Reagent to stop reaction and deplete remaining ATP. Incubate 40 min. Add 10 µL of Kinase Detection Reagent to convert ADP to ATP and generate luminescence. Incubate 30 min.
- Readout: Measure luminescence on a plate reader (e.g., PerkinElmer EnVision).
- Data Analysis: Calculate % inhibition:
(1 - (Cpdsignal - LowCtrl)/(HighCtrl - LowCtrl)) * 100. Compounds with >70% inhibition proceed to IC50 determination in dose-response.
Title: HTS Workflow for Kinase Inhibitor Screening
Application Note: Mechanistic Study of Inhibition Mode
Objective: Determine the mechanism of action (competitive, non-competitive, uncompetitive) of a validated hit against a protease target. Thesis Context: The Cheng-Prusoff derivation assumes competitive inhibition. Mechanistic studies are required to verify this assumption. Ki values derived under correct mechanistic models are essential for meaningful in vitro-in vivo correlations.
Key Quantitative Data (Representative): Table 3: Enzyme Kinetic Data for Compound X
| [Inhibitor] | Apparent Km (µM) | Apparent Vmax (RFU/min) | Inferred Mechanism |
|---|---|---|---|
| 0 (DMSO) | 25.0 ± 1.5 | 12000 ± 300 | Baseline |
| 0.5 µM | 50.1 ± 3.2 | 11800 ± 250 | Competitive |
| 1.0 µM | 75.8 ± 4.1 | 11500 ± 400 | Competitive |
| 2.0 µM | 124.5 ± 8.7 | 11900 ± 500 | Competitive |
Protocol 2.2.1: Determining Inhibition Mode via Steady-State Kinetics
- Substrate Titration: Prepare a 2X serial dilution of substrate (peptide) in assay buffer, covering 0.2xKm to 5xKm (e.g., 5 to 100 µM).
- Inhibitor Dilution: Prepare 3 concentrations of inhibitor (e.g., 0.5x, 1x, 2x estimated Ki) and a vehicle (DMSO) control.
- Reaction Setup: In a 96-well plate, mix 25 µL of substrate solution with 25 µL of inhibitor solution. Start reaction by adding 50 µL of enzyme solution.
- Initial Rate Measurement: Monitor product formation continuously (e.g., fluorescence increase) for 10-15 minutes using a plate reader. Record the linear slope (initial velocity, V0) for each well.
- Data Fitting: Fit the data for each inhibitor concentration to the Michaelis-Menten equation to obtain apparent Km and Vmax. Graphically analyze using Lineweaver-Burk (1/V vs 1/[S]) or Dixon plots.
- Ki Calculation: For confirmed competitive inhibition, perform global fit of all data to the competitive inhibition model using software (e.g., Prism, GraphPad) to extract the true Ki.
Title: Mechanistic Study Workflow for Inhibition Mode
Critical Data Conversion Table
Table 4: Cheng-Prusoff Correction in Practice
| Application Stage | Typical IC50 Range | [S]/Km Ratio | Correction Factor (1+[S]/Km) | Impact on Ki vs IC50 |
|---|---|---|---|---|
| Primary HTS | 0.1 - 10 µM | 0.67 (10µM/15µM) | 1.67 | Ki ≈ 0.6 x IC50 (Potency overestimated by HTS IC50) |
| SAR Optimization | 1 nM - 1 µM | 1.0 (at Km) | 2.0 | Ki = 0.5 x IC50 (Critical for comparing analogs) |
| Mechanistic Studies | Near Ki | Varied (0.1-5) | 1.1 - 6.0 | Ki significantly lower if [S] > Km. Validates CP assumptions. |
Beyond the Basics: Troubleshooting Common Pitfalls and Optimizing Accuracy
Within the thesis context of advancing the Cheng-Prusoff equation for IC50 to Ki conversion, this document addresses critical deviations from ideal behavior in competitive binding assays. The Cheng-Prusoff derivation assumes: 1) equilibrium conditions, 2) ligand depletion is negligible, 3) radioligand binding is to a single, non-interacting site, 4) the inhibitor is purely competitive, and 5) no allosteric or off-target effects occur. Violations of these assumptions lead to systematic errors in Ki estimation, impacting drug discovery.
Table 1: Assumptions of the Cheng-Prusoff Equation and Indicators of Failure
| Assumption | Condition for Validity | Common Breakdown | Impact on Ki Estimate |
|---|---|---|---|
| Equilibrium | Incubation time >> ligand Koff-1 | Slow inhibitor kinetics; insufficient incubation. | Under- or overestimation depending on direction. |
| No Ligand Depletion | [Radioligand] >> Kd and [RT] very low. | High receptor concentration or low radioligand affinity. | Ki estimate is overestimated. |
| Single Site | Radioligand binds one homogeneous population. | Binding to multiple sites or receptor isoforms. | Non-linear Schild plot; IC50 dependent on [L]. |
| Purely Competitive | Inhibitor and radioligand are mutually exclusive. | Allosteric inhibition or binding to an unrelated site. | IC50 does not shift as predicted with [L]; plateau <100%. |
| No Off-Target Effects | Inhibitor is specific for the target receptor. | Binding to transporters, enzymes, or other receptors. | Discrepancy between binding and functional assays. |
Table 2: Experimental Red Flags and Diagnostic Tests
| Observed Anomaly | Diagnostic Experiment | Interpretation of Positive Result |
|---|---|---|
| IC50 not proportional to [L] | Vary [radioligand] over 10-100x Kd. | Violation of competitive model (allostery, ligand depletion). |
| Hill coefficient ≠ 1 | Fit data to 4-parameter logistic equation. | Suggests multiple binding sites or cooperativity. |
| Incomplete inhibition | Test inhibitor at very high conc. (e.g., 100 µM). | Suggests non-competitive component or off-target binding. |
| Ki(binding) ≠ Ki(functional) | Compare in orthogonal functional assay (e.g., cAMP, Ca2+). | Suggests off-target or signaling pathway effects. |
Detailed Experimental Protocols
Protocol 1: Diagnosing Ligand Depletion and Non-Specific Binding
Objective: To correct measured IC50 for ligand depletion and validate the no-depletion assumption. Materials: See "Scientist's Toolkit" (Section 5). Workflow:
- Perform a saturation binding experiment to determine the Kd and Bmax of the radioligand.
- Calculate the fractional receptor occupancy (f) in your inhibition assay: f = [RT] / (Kd + [L]), where [L] is free radioligand concentration.
- If f > 0.1, ligand depletion is significant. The corrected free concentration is: [L]free = [L]total - Bound.
- Re-fit inhibition data using [L]free in the Cheng-Prusoff correction: Ki = IC50 / (1 + [L]free/Kd).
- Validate by repeating assay with 10-fold lower membrane protein concentration; if IC50 shifts, depletion was a factor.
Protocol 2: Testing for Allosteric vs. Orthosteric Inhibition
Objective: To determine if an inhibitor is competitive or allosteric. Workflow:
- Conduct competitive inhibition assays across a minimum of three different radioligand concentrations (e.g., 0.3x Kd, 1x Kd, 3x Kd).
- Plot IC50 values against [L]/Kdpure competitive inhibitor, the slope of IC50 vs. [L] will be 1.
- For an allosteric inhibitor, the plot will be non-linear, and a full curve must be globally fit to an allosteric model to derive an affinity (KB) and a cooperativity factor (α).
- Perform dissociation kinetic experiments: Pre-bind radioligand to receptor, then dilute and add excess unlabeled competitor. A competitive inhibitor will accelerate radioligand dissociation, while an allosteric inhibitor may decelerate or not affect it.
Protocol 3: Identifying Off-Target Effects via Selectivity Panel Screening
Objective: To assess inhibitor specificity across related and unrelated targets. Workflow:
- Select a panel of 50-100 related receptors, enzymes, ion channels, and transporters relevant to the therapeutic area.
- Run primary binding or functional assays for each panel target at a single, high concentration of the inhibitor (e.g., 10 µM).
- For targets showing >50% inhibition at 10 µM, run a full concentration-response curve to determine IC50 and Ki values.
- Calculate selectivity ratios (Ki(primary target) / Ki(off-target)). A ratio >100 is typically desirable for a selective tool compound.
Visualization Diagrams
Diagram Title: Assumption Breakdown Leading to Inaccurate Ki
Diagram Title: Diagnostic Flowchart for IC50 to Ki Validation
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents for Investigating Non-Ideal Behavior
| Item | Function & Rationale |
|---|---|
| Membrane Preparation from overexpressing cell lines | Provides a consistent, enriched source of the target receptor for binding studies. High Bmax can exacerbate ligand depletion, making it a useful stress test. |
| High-Specific Activity Radioligand (e.g., [³H], [¹²⁵I]) | Enables work at low nanomolar concentrations to minimize ligand depletion and non-specific binding, adhering to ideal assay conditions. |
| Reference Orthosteric Antagonist (cold ligand) | Essential positive control for validating assay performance and competitive inhibition kinetics. |
| Reference Allosteric Modulator | Critical control for distinguishing allosteric vs. orthosteric mechanisms in dissociation kinetics experiments. |
| Scintillation Proximity Assay (SPA) Beads | Allows for homogeneous, no-wash binding assays, reducing manipulation errors and enabling higher throughput for [L] shift experiments. |
| β-Arrestin or Second Messenger Assay Kits | Orthogonal functional assays (e.g., cAMP, Ca²⁺, BRET) to compare binding Ki with functional Ki and detect signaling bias or off-target effects. |
| Selectivity Panel Screening Service | Access to curated panels of unrelated targets (GPCRs, kinases, ion channels) is crucial for systematic off-target profiling. |
| Software for Global Fitting (e.g., Prism, GraphPad) | Required for fitting data to complex models (allosteric, two-site) to derive accurate parameters when simple Cheng-Prusoff fails. |
This application note addresses a critical, often overlooked, source of systematic error within the broader thesis on the rigorous application of the Cheng-Prusoff equation for converting half-maximal inhibitory concentration (IC₅₀) to inhibition constant (Kᵢ) values in drug discovery research. The Cheng-Prusoff derivation assumes accurate knowledge of the dissociation constant (Kd) of the labeled ligand and the Michaelis constant (Km) of the substrate, as well as precise control and measurement of their concentrations ([L] and [S]). Inaccurate determination of these parameters directly propagates into significant errors in the reported Kᵢ, leading to misinformed structure-activity relationships (SAR), flawed compound prioritization, and ultimately, costly failures in downstream development.
Quantifying the Impact: Error Propagation Analysis
Errors in Km, Kd, [S], and [L] are not simply additive; they propagate non-linearly through the Cheng-Prusoff equations. The tables below summarize the quantitative impact of common measurement inaccuracies on calculated Kᵢ values for both competitive and non-competitive inhibition models.
Table 1: Impact of Parameter Errors on Calculated Kᵢ (Competitive Inhibition) Equation: Kᵢ = IC₅₀ / (1 + [S]/Km) for enzymes; Kᵢ = IC₅₀ / (1 + [L]/Kd) for receptors
| Parameter | True Value | Measured/Used Value | % Error in Parameter | Resultant % Error in Kᵢ (Example: IC₅₀=1 µM, [S] or [L]=Km or Kd) |
|---|---|---|---|---|
| Km or Kd | 10 µM | 5 µM | -50% | +100% (Kᵢ is 2-fold overestimated) |
| Km or Kd | 10 µM | 20 µM | +100% | -33% (Kᵢ is 1.5-fold underestimated) |
| [S] or [L] | 10 µM (≈Km/Kd) | 5 µM | -50% | -33% (Kᵢ is 1.5-fold underestimated) |
| [S] or [L] | 10 µM (≈Km/Kd) | 20 µM | +100% | +100% (Kᵢ is 2-fold overestimated) |
Table 2: Magnification of Error at High [S]/Km or [L]/Kd Ratios Error is magnified when substrate or ligand concentration is high relative to its binding constant.
| Assay Condition ([S]/Km or [L]/Kd ratio) | True Kᵢ (nM) | If Km or Kd is 50% Underestimated, Calculated Kᵢ Becomes: | Fold Error |
|---|---|---|---|
| Low ([S]=Km) | 10 | 20 nM | 2.0 |
| High ([S]=10*Km) | 10 | 110 nM | 11.0 |
Experimental Protocols for Accurate Parameter Determination
Protocol 3.1: Determination of Km via Initial Rate Kinetics
Objective: To accurately determine the Michaelis constant (Km) for an enzymatic assay used in IC₅₀ generation.
Key Reagents & Materials: Purified enzyme, synthetic substrate, reaction buffer, detection system (fluorogenic/chromogenic), multi-well plate reader.
Procedure:
- Prepare a 2X serial dilution of substrate across 10-12 concentrations, spanning from ~0.2× to 5× the estimated Km.
- In a 96-well plate, mix equal volumes of enzyme solution (at a fixed, low concentration) and each substrate dilution to initiate the reaction. Perform in triplicate.
- Monitor product formation (e.g., absorbance, fluorescence) continuously for 10-15 minutes to obtain initial velocities (v₀).
- Fit the collected data (v₀ vs. [S]) to the Michaelis-Menten equation using non-linear regression software:
v₀ = (Vmax * [S]) / (Km + [S]). - Validation: Ensure the fitted curve has a clear hyperbolic shape. Report Km with 95% confidence intervals from the fit.
Protocol 3.2: Saturation Binding for Kd Determination
Objective: To accurately determine the equilibrium dissociation constant (Kd) of a radiolabeled or fluorescent ligand for a receptor binding assay.
Key Reagents & Materials: Membrane preparation or purified receptor, labeled ligand (hot), unlabeled ligand (cold for NSB), binding buffer, filtration harvester or proximity assay plate.
Procedure:
- Prepare a dilution series of the labeled ligand, typically spanning from ~0.1× to 10× the estimated Kd.
- For each concentration, set up total binding (TB) wells (receptor + labeled ligand) and nonspecific binding (NSB) wells (receptor + labeled ligand + excess unlabeled competitor).
- Incubate to equilibrium (time determined in separate kinetic experiment).
- Separate bound from free ligand (via filtration or other means) and quantify bound signal.
- Calculate specific binding (SB) = TB - NSB for each point.
- Fit SB vs. [L] to a one-site specific binding model:
SB = (Bmax * [L]) / (Kd + [L]). - Validation: The Hill slope should be ~1.0. The Kd should be consistent with the concentration at which binding is half of Bmax.
Protocol 3.3: Accurate Quantification of Stock [S] and [L]
Objective: To minimize errors in the working concentration of substrates and ligands.
Procedure:
- Quantitative Analysis: Use validated methods (NMR, quantitative amino acid analysis, UV-Vis spectroscopy using known extinction coefficients) to determine the exact concentration of stock solutions, accounting for purity, salt form, and water content.
- Serial Dilution Discipline: Use calibrated pipettes and perform serial dilutions in a logical, minimal-step sequence. Use the same buffer for dilutions as in the assay to avoid adsorption losses.
- Documentation: Record lot numbers, measured stock concentrations, and all dilution factors.
Visualizing the Error Propagation Pathway
Title: Error Propagation from Inputs to Kᵢ and Impact
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Reliable Kᵢ Conversion
| Item | Function & Importance for Accurate Kᵢ |
|---|---|
| High-Purity, Quantified Substrate/Ligand | Foundation for accurate [S] and [L]. Must be analytically quantified (NMR, qNMR, elemental analysis) to know exact concentration, not just mass. |
| Validated Kd/Km Reference Compound | A well-characterized inhibitor or ligand with a literature Kd/Km. Serves as a crucial internal control to validate the entire assay system and parameter set. |
| Stable, Purified Enzyme/Receptor Prep | Batch-to-batch consistency in protein activity and concentration is vital for reproducible Km/Kd measurements. Use aliquots from a single large prep for a project. |
| Calibrated Liquid Handling Systems | Especially for serial dilutions. Regular calibration of pipettes and use of positive displacement pipettes for viscous solutions reduces [S]/[L] preparation errors. |
| Radiolabeled Ligand with Known SA | For binding assays, specific activity (SA) must be high and precisely known from the supplier to calculate [L] accurately from counts. |
| Non-Linear Regression Software | Essential for robust fitting of Michaelis-Menten and binding isotherm data to extract Km/Kd with confidence intervals (e.g., GraphPad Prism, SCIENTIST). |
| Low-Binding Labware | Polypropylene plates/tubes minimize adsorption losses of precious substrates, ligands, and inhibitors, preserving intended concentrations. |
Within the canonical framework of enzyme inhibition kinetics, the Cheng-Prusoff equation provides a vital bridge for converting experimentally measured IC₅₀ values to the fundamental binding constant, Kᵢ. This conversion relies on the central assumption that the total inhibitor concentration ([I]ₜₒₜ) is in vast excess over the total enzyme concentration ([E]ₒₜₐₗ), i.e., [I]ₜₒₜ ≈ [I]fᵣₑₑ. This condition ensures the free inhibitor concentration is not significantly depleted by binding. However, for tight-binding inhibitors—characterized by Kᵢ values comparable to or lower than [E]ₒₜₐ₹—this assumption collapses. Under tight-binding conditions ([I] ≈ [E]), the observed IC₅₀ is significantly elevated and becomes dependent on [E]ₒₜₐ₹, leading to a substantial underestimation of inhibitor potency if the standard Cheng-Prusoff correction is applied naively. This application note, framed within a broader thesis on the limitations and proper application of the Cheng-Prusoff paradigm, details the identification, analysis, and accurate Kᵢ determination for tight-binding inhibitors.
Key Concepts and Quantitative Data
Table 1: Impact of Tight-Binding Conditions on Observed IC₅₀
| Condition | Assumption | Relationship between IC₅₀ and Kᵢ | Consequence of Standard Cheng-Prusoff |
|---|---|---|---|
| Classical ([I]ₜ >> [E]ₒ) | [I]fᵣₑₑ ≈ [I]ₜ | IC₅₀ = Kᵢ(1 + [S]/Kₘ) | Accurate Kᵢ derivation. |
| Tight-Binding ([I]ₜ ≈ [E]ₒ) | Significant [I] depletion | IC₅₀ = Kᵢ(1 + [S]/Kₘ) + [E]ₒ/2 | IC₅₀ is inflated; Kᵢ is severely underestimated. |
Table 2: Comparison of Kᵢ Determination Methods for Tight-Binding Inhibitors
| Method | Principle | Key Requirements/Advantages | Typical Output |
|---|---|---|---|
| Morrison Quadratic | Solves exact equilibrium equation. | Requires known [E]ₒₜₐₗ; Accounts for depletion. | Accurate Kᵢ, even when IC₅₀ ≈ [E]ₒ/2. |
| IC₅₀ vs. [E]ₒ Plot | Linear fit of IC₅₀ = [E]ₒ/2 + Kᵢ(1+[S]/Kₘ). | Multiple experiments at varying [E]ₒₜₐₗ. | Slope yields Kᵢ(app); y-intercept gives Kᵢ. |
| Progress Curve Analysis | Models time-dependent product formation under inhibition. | Continuous assay; Can also characterize slow-binding kinetics. | Direct fit for kₒₙ, kₒff, and Kᵢ*. |
Experimental Protocols
Protocol 1: Identifying Tight-Binding Behavior via [E]ₜ Dependency Test
Objective: To determine if an inhibitor exhibits tight-binding characteristics by measuring IC₅₀ at multiple enzyme concentrations. Materials: See "The Scientist's Toolkit" below. Procedure:
- Prepare a dilution series of the inhibitor (e.g., 3-fold dilutions covering a range above and below the suspected Kᵢ).
- Set up enzymatic reactions in triplicate with a fixed, saturating (for competitive inhibitors) or low (for other types) substrate concentration.
- Crucial Step: Perform the entire dose-response experiment using at least three different, accurately quantified total enzyme concentrations (e.g., 0.1 nM, 0.5 nM, and 2.0 nM).
- Measure initial reaction velocities (vᵢ) for each [I] and [E]ₒ condition.
- For each [E]ₒ dataset, fit the dose-response curve (vᵢ/v₀ vs. log[I]ₜ) to a 4-parameter logistic model to determine the IC₅₀.
- Analysis: Plot the observed IC₅₀ values as a function of total enzyme concentration ([E]ₒ). A positive, linear correlation is diagnostic of tight-binding inhibition.
Protocol 2: Determining Accurate Kᵢ Using the Morrison Quadratic Equation
Objective: To calculate the true Kᵢ from a single dose-response curve under tight-binding conditions. Procedure:
- Conduct a detailed dose-response experiment as in Protocol 1, using a single, well-quantified [E]ₒ that is comparable to the suspected Kᵢ.
- Determine the uninhibited velocity (v₀) and the velocity at each inhibitor concentration (vᵢ).
- Calculate fractional activity (vᵢ/v₀) for each [I]ₜ.
- Fit the data using the Morrison equation for competitive inhibition:
vᵢ/v₀ = 1 - (([E]ₒ + [I]ₜ + Kᵢ*(1+[S]/Kₘ)) - sqrt(([E]ₒ + [I]ₜ + Kᵢ*(1+[S]/Kₘ))² - 4*[E]ₒ*[I]ₜ)) / (2*[E]ₒ) - In the fit, fix the experimentally determined values for [E]ₒ, [S], and Kₘ. The primary fitted parameter is Kᵢ. Note: This equation is implemented in software like GraphPad Prism ("Competitive inhibition -- tight binding" equation) or can be solved via non-linear regression in other platforms.
Visualizations
Decision Workflow for Tight-Binding Inhibitor Analysis
Equilibrium in Tight-Binding Inhibition
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Tight-Binding Studies |
|---|---|
| High-Precision Enzymes (≥95% purity) | Accurate quantification of active [E]ₒₜₐₗ is non-negotiable. Impurities lead to significant errors in Kᵢ calculation. |
| Substrate with Known Kₘ | The Michaelis constant must be precisely determined under assay conditions for correct Kᵢ(app) correction. |
| Low-Binding Microplates & Tips | Minimizes non-specific adsorption of potent inhibitors, which can distort dose-response curves. |
| High-Resolution DMSO Stocks | Use anhydrous DMSO and gravimetric preparation for exact, reproducible inhibitor stock concentrations. |
| Continuous Activity Assay Reagents | Essential for progress curve analysis (e.g., coupled spectrophotometric/fluorogenic assays). |
| Software for Non-Linear Regression | Programs capable of fitting data to the Morrison quadratic equation (e.g., GraphPad Prism, KinTek Explorer). |
| Active Site Titrant | A known, tight-binding standard inhibitor to determine the active concentration of the enzyme stock. |
1. Introduction & Thesis Context Within the framework of advanced research on the Cheng-Prusoff equation and its application in converting half-maximal inhibitory concentration (IC₅₀) to dissociation constant for inhibitor (Kᵢ), assay design is paramount. The foundational equation, Kᵢ = IC₅₀ / (1 + [S]/Kₘ + [L]/Kd), highlights the critical dependence of the derived Kᵢ on the concentrations of substrate ([S]) and competing ligand ([L]). Incorrect selection of [S] and [L] leads to significant errors in Kᵢ estimation, undermining drug discovery efforts. This application note provides protocols and design principles for optimizing these key parameters to ensure robust and accurate Kᵢ determination.
2. Core Principles for [S] and [L] Selection The goal is to design conditions where the IC₅₀ value is a reasonable approximation of the Kᵢ, minimizing the correction factor from the Cheng-Prusoff equation. This enhances assay sensitivity and reliability.
- Substrate Concentration ([S]): Must be carefully set relative to its Michaelis constant (Kₘ).
- Competing Ligand Concentration ([L]): Must be carefully set relative to its dissociation constant (Kd).
Table 1: Guidelines for Selecting [S] and [L] Concentrations
| Parameter | Optimal Range | Rationale | Impact of Deviation |
|---|---|---|---|
| [S] / Kₘ Ratio | 0.1 – 1.0 (Ideal: ≤ 1) | Minimizes the (1 + [S]/Kₘ) term, reducing its influence on Kᵢ calculation. A ratio of 1 yields a 2-fold correction. | High Ratio (>5): IC₅₀ >> Kᵢ, large correction required, assay insensitive to tight binders. Very Low Ratio: May compromise signal-to-noise. |
| [L] / Kd Ratio | As low as feasibly possible (Ideal: << 1) | Minimizes the ([L]/Kd) term. For radioligand binding, [L] is often set at ~Kd to balance signal and correction. | High Ratio (>1): IC₅₀ >> Kᵢ, large correction, increased error propagation from Kd uncertainty. |
| Target Correction Factor (1 + [S]/Kₘ + [L]/Kd) | 2 – 10 | A manageable correction factor that provides sufficient signal while avoiding excessive inflation of IC₅₀. | Very Large Factor: IC₅₀ is a poor estimator of Kᵢ, magnifying errors in Kₘ/Kd. Factor ~1: Ideal but often experimentally impractical. |
3. Experimental Protocols
Protocol A: Determining Kₘ for Enzyme Assay Substrate Optimization Objective: Accurately determine the Michaelis constant (Kₘ) to inform [S] selection. Materials: See "Scientist's Toolkit" (Table 3). Procedure:
- Prepare a serial dilution of the substrate ([S]) across a broad range (e.g., 0.1× to 10× the estimated Kₘ).
- In a reaction plate, combine fixed concentrations of enzyme (well below [S]) and assay buffer with varying [S] in triplicate.
- Initiate the reaction by adding a cofactor or second substrate as required. Include negative controls (no enzyme).
- Monitor product formation kinetically using an appropriate detection method (e.g., absorbance, fluorescence).
- Calculate initial velocity (V₀) for each [S] from the linear phase of the progress curve.
- Fit the V₀ vs. [S] data to the Michaelis-Menten model (V₀ = (Vₘₐₓ * [S]) / (Kₘ + [S])) using non-linear regression software to extract Kₘ and Vₘₐₓ.
Protocol B: Determining Kd for Ligand Binding Assay Optimization Objective: Accurately determine the equilibrium dissociation constant (Kd) for the competing ligand ([L]). Materials: See "Scientist's Toolkit" (Table 3). Procedure:
- Prepare a serial dilution of the unlabeled (cold) competing ligand or a different radiolabeled/fluorescent tracer ligand.
- In a binding plate, combine a fixed, low concentration of the labeled ligand ([L*]), membrane preparation or purified receptor, and assay buffer with varying concentrations of the competing/tracer ligand. Include total binding (no competitor) and non-specific binding (NSB, with a large excess of unlabeled competitor) controls.
- Incubate to equilibrium (time determined empirically).
- Separate bound from free ligand via filtration, centrifugation, or other appropriate method.
- Quantify bound labeled ligand (e.g., scintillation counting, fluorescence).
- Subtract NSB from all values to obtain specific binding.
- Fit the specific binding vs. ligand concentration data to a one-site specific binding model to derive Kd and Bₘₐₓ.
Protocol C: IC₅₀ Shift Assay to Validate Cheng-Prusoff Conditions Objective: Confirm the appropriate relationship between [S]/Kₘ or [L]/Kd by observing the predicted shift in IC₅₀. Materials: See "Scientist's Toolkit" (Table 3). Procedure:
- For an enzyme assay: Perform inhibitor dose-response curves at two different substrate concentrations (e.g., [S] = Kₘ and [S] = 0.2 * Kₘ).
- For a binding assay: Perform inhibitor dose-response curves at two different ligand concentrations (e.g., [L] = Kd and [L] = 0.3 * Kd).
- Determine the IC₅₀ for the inhibitor under each condition via 4-parameter logistic fit.
- Compare the IC₅₀ values. The IC₅₀ at the higher [S] or [L] should be predictably higher. Use the Cheng-Prusoff equation with the known Kₘ or Kd to check if the shift aligns with theoretical prediction.
4. Visualization of Concepts and Workflows
Title: Assay Optimization Workflow for Accurate Ki Determination
Title: Relationship of Assay Parameters to Ki via Cheng-Prusoff
5. The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions & Materials
| Item | Function/Explanation |
|---|---|
| Purified Enzyme / Receptor Preparation | The biological target. Source (recombinant, tissue) and purity must be consistent for reproducible Kₘ/Kd and IC₅₀ determinations. |
| Characterized Substrate | For enzyme assays. Must have a known detection method (chromogenic, fluorogenic) and stable kinetic properties. |
| High-Affinity Radioligand or Fluorescent Tracer | For binding assays. Must have high specific activity/brightness, low non-specific binding, and a known Kd. |
| Reference Inhibitor (Control Compound) | A well-characterized inhibitor with known potency (Kᵢ) for validating assay performance and Cheng-Prusoff conversion. |
| Assay Buffer with Cofactors/Mg²⁺ | Maintains optimal pH, ionic strength, and provides essential components for enzyme/ligand binding activity. |
| Detection System | Plate reader (absorbance, fluorescence, luminescence) or scintillation counter. Must be sensitive and stable. |
| Nonlinear Regression Analysis Software | Essential for robust fitting of data to Michaelis-Menten, binding isotherm, and dose-response models (e.g., GraphPad Prism). |
| Liquid Handling System | For accurate and reproducible serial dilutions of substrates, ligands, and inhibitors, minimizing human error. |
The Cheng-Prusoff equation is a cornerstone in receptor pharmacology and drug discovery, providing a mathematical relationship to convert the experimentally measured half-maximal inhibitory concentration (IC₅₀) to the equilibrium inhibition constant (Kᵢ). This conversion is critical as Kᵢ represents a true thermodynamic constant, independent of assay conditions like substrate or ligand concentration. A core challenge, often overlooked, is the statistically rigorous propagation of error associated with the IC₅₀ measurement through the Cheng-Prusoff equation to yield a confidence interval for the final Kᵢ. This article details protocols and application notes for accurately performing this error propagation, a vital step for robust quantitative comparison of compound potency.
Theoretical Framework & Error Propagation Formula
The standard Cheng-Prusoff correction for competitive inhibitors is: Kᵢ = IC₅₀ / (1 + [S]/Kₘ) (for enzyme assays) Kᵢ = IC₅₀ / (1 + [L]/K₅₀) (for receptor binding assays)
Where:
- [S]: Concentration of substrate.
- Kₘ: Michaelis constant for the substrate.
- [L]: Concentration of the competing radioligand.
- K₅₀: Dissociation constant of the radioligand.
Each variable (IC₅₀, [S], Kₘ) carries its own experimental error (typically Standard Error, SE, or standard deviation, SD). The propagated variance (Var) for Kᵢ can be approximated using the first-order Taylor expansion (error propagation formula):
Var(Kᵢ) ≈ (δKᵢ/δIC₅₀)² * Var(IC₅₀) + (δKᵢ/δ[S])² * Var([S]) + (δKᵢ/δKₘ)² * Var(Kₘ)
Assuming [S] is known with high precision (negligible error), the formula simplifies to: Var(Kᵢ) ≈ [Var(IC₅₀) + (IC₅₀²/Kₘ⁴) * Var(Kₘ)] / (1 + [S]/Kₘ)⁴
The Standard Error of Kᵢ is then: SE(Kᵢ) = √[Var(Kᵢ)] The 95% Confidence Interval (CI) for Kᵢ is: Kᵢ ± t_(df, 0.975) * SE(Kᵢ), where t is the Student's t-value for the appropriate degrees of freedom (df).
Table 1: Example Data Set for Error Propagation Calculation
| Parameter | Symbol | Value | Standard Error (SE) | Notes |
|---|---|---|---|---|
| Measured IC₅₀ | IC₅₀ | 100 nM | ± 15 nM | From 4-parameter logistic fit (n=3 independent experiments) |
| Substrate Concentration | [S] | 10 µM | ± 0.1 µM | Assumed precisely known from stock preparation |
| Michaelis Constant | Kₘ | 5 µM | ± 0.7 µM | Determined from separate kinetic assay (n=6) |
| Calculated Kᵢ | Kᵢ | 33.3 nM | ± 6.2 nM | Propagated using simplified formula |
| 95% CI for Kᵢ | CI | 33.3 ± 16.2 nM | df=4, t=2.776 |
Table 2: Impact of [S]/Kₘ Ratio on Error Magnification
| [S] / Kₘ Ratio | Correction Factor (1+[S]/Kₘ) | Kᵢ (if IC₅₀=100nM) | Approx. Error Magnification Factor* |
|---|---|---|---|
| 0.2 (Subsaturing) | 1.2 | 83.3 nM | ~1.2x |
| 1.0 (At Kₘ) | 2.0 | 50.0 nM | ~2.0x |
| 5.0 (High) | 6.0 | 16.7 nM | ~6.0x |
*Relative magnification of the relative error in IC₅₀ on the relative error in Kᵢ. High ratios amplify error dramatically.
Experimental Protocols
Protocol 4.1: Determining IC₅₀ with Reliable Error Estimation
Objective: To generate a dose-response curve and calculate an IC₅₀ value with a robust standard error. Materials: Target enzyme/receptor, substrate/ligand, inhibitor compound, assay buffer, detection reagents (e.g., fluorescent, luminescent). Procedure:
- Perform the inhibitory activity assay in triplicate, across a minimum of 10 inhibitor concentrations (e.g., 0.1 nM to 100 µM, serial dilutions).
- Conduct a minimum of three independent experiments on separate days with fresh reagent preparations.
- For each experiment, fit the dose-response data (response vs. log[Inhibitor]) to a 4-parameter logistic (4PL) model using robust non-linear regression software (e.g., GraphPad Prism).
- Record the best-fit IC₅₀ and its standard error (SE_IC50) or confidence interval from each experiment.
- Do not simply pool all data points from all runs into a single curve fit. Instead, calculate the global mean IC₅₀ and its standard error of the mean (SEM) from the n independent IC₅₀ estimates. SEM_IC50 = SD(IC₅₀ estimates) / √n.
Protocol 4.2: Determining Kₘ or K₅₀ with Error
Objective: To accurately determine the Michaelis (Kₘ) or ligand dissociation (K₅₀) constant and its variance. A. For Enzymatic Kₘ:
- Measure initial reaction velocities (v₀) over a range of substrate concentrations [S] (e.g., 0.2Kₘ to 5Kₘ).
- Perform in duplicate across ≥3 independent experiments.
- Fit the data from each experiment to the Michaelis-Menten model (v₀ = (Vₘₐₓ*[S])/(Kₘ+[S])) via non-linear regression.
- Report the mean Kₘ and its SEM from the independent estimates.
B. For Receptor Binding K₅₀:
- Perform saturation binding with a range of radioligand concentrations.
- Perform in duplicate across ≥3 independent experiments.
- Fit specific binding data to a one-site specific binding model (B = (Bₘₐₓ*[L])/(K₅₀+[L])).
- Report the mean K₅₀ and its SEM.
Protocol 4.3: Propagating Error to Kᵢ
Objective: To calculate the final Kᵢ value with its 95% confidence interval. Materials: IC₅₀ ± SEM_IC50; Kₘ or K₅₀ ± SEM; [S] or [L] value; statistical software or calculator. Procedure:
- Calculate the mean Kᵢ using the mean IC₅₀, mean Kₘ/K₅₀, and the known [S]/[L] in the Cheng-Prusoff equation.
- Apply the simplified error propagation formula (assuming negligible error in [S]/[L]):
- Calculate Var(IC₅₀) = (SEMIC50)²
- Calculate Var(Kₘ) = (SEMKₘ)²
- Compute Var(Kᵢ) using the formula in Section 2.
- Compute SE_Ki = √[Var(Ki)]
- Determine degrees of freedom (df). A conservative estimate is df = n_IC50 + n_Km - 2, where n is the number of independent experiments for each parameter.
- Find the two-tailed t-value (t* ) for df at α=0.05 (e.g., from t-distribution table).
- Calculate: 95% CI for Kᵢ = Kᵢ ± (t* × SE_Ki).
- Report as: Kᵢ = 33.3 nM (95% CI: 17.1 to 49.5 nM).
Visualizations
Title: Workflow for Propagating Error to Ki
Title: Key Equations for Error Propagation
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Solutions for Reliable Ki Determination
| Item | Function / Relevance | Critical Note for Error Control |
|---|---|---|
| High-Purity Target Protein | Enzyme or receptor for assay. Source variability affects Kₘ/K₅₀. | Use consistent source/purification batch across all experiments (IC₅₀ & Kₘ). |
| Validated Substrate/Radioligand | The competing molecule in the Cheng-Prusoff equation. | Precisely determine concentration ([S] or [L]) via spectrophotometry/quantitation. Minimize its error. |
| Reference Inhibitor (Control) | Compound with well-established Kᵢ. Validates assay conversion. | Use to verify the accuracy of the entire IC₅₀→Kᵢ workflow, including error bounds. |
| Assay Plate Reader (Kinetic Capable) | For measuring reaction velocity or binding signal over time. | Instrument stability and linear detection range are crucial for robust IC₅₀ and Kₘ curves. |
| Statistical Software (e.g., GraphPad Prism) | For non-linear regression of dose-response & saturation data, and error propagation. | Must be capable of reporting parameter standard errors/CIs from curve fits and performing custom error calculations. |
| Liquid Handling System | For serial dilution of inhibitors and reagent dispensing. | Reduces technical error in concentration preparation, a key source of variance in IC₅₀. |
Accurate determination of the inhibition constant (Ki) from the half-maximal inhibitory concentration (IC50) via the Cheng-Prusoff equation is foundational to enzymology and drug discovery. The validity of this conversion rests upon strict assumptions, including the steady-state condition, negligible substrate depletion, stable enzyme activity, and a robust signal-to-noise ratio. Deviations from these assumptions introduce systematic errors, leading to inaccurate Ki estimates and misleading structure-activity relationships. This Application Note provides detailed protocols and analyses for identifying and correcting these critical experimental artifacts.
Substrate Depletion: Quantification and Correction
Substrate concentration ([S]) must remain close to the initial concentration ([S]0) throughout the reaction. Significant depletion (>10%) violates the Michaelis-Menten condition [S] >> [E], causing IC50 to underestimate true Ki.
Protocol 1.1: Assessing Substrate Depletion
Objective: Quantify the percentage of substrate consumed under assay conditions. Materials:
- Reaction mix (enzyme, buffer, cofactors)
- Substrate stock at Km concentration
- Stopping reagent (e.g., acid, EDTA, inhibitor)
- Analytical method (LC-MS, fluorescence calibration curve)
Procedure:
- Set up kinetic reactions in triplicate at your standard assay conditions ([S] = Km).
- At time = 0 (immediately after mixing), quench one set of reactions.
- Quench remaining reactions at the standard assay endpoint time (t_end).
- Quantify the amount of product formed or substrate remaining using a calibrated absolute quantification method (e.g., LC-MS against a standard curve, or fluorescence correlated to a product standard).
- Calculate % Depletion:
( [P]_t_end / [S]_0 ) * 100.
Interpretation: If depletion >10%, corrections are mandatory.
Correction Strategy:
- Increase [S]0: Use [S] ≥ 5*Km to ensure ≤10% consumption. This may require optimizing detection sensitivity.
- Shorten Reaction Time: Use initial velocity conditions (≤5-10% conversion).
- Numerical Correction: Apply the Morrison equation for tight-binding inhibitors or integrated rate equations when [S] depletion is significant.
Table 1: Impact of Substrate Depletion on IC50/Ki Discrepancy
| % Substrate Depletion | [S]/Km Ratio | Observed IC50 / True Ki (for Competitive Inhibitor) | Recommended Action |
|---|---|---|---|
| ≤5% | ≥10 | ~1.0 | None. Assumption valid. |
| 10% | 5 | ~0.85 | Borderline. Increase [S] or reduce time. |
| 20% | 2.5 | ~0.70 | Significant error. Must correct conditions. |
| 50% | 1 | ~0.50 | Severe error. Data invalid for Cheng-Prusoff. |
Enzyme Instability: Monitoring and Normalization
Enzyme activity loss during the assay (e.g., due to thermal denaturation, proteolysis) leads to an overestimation of inhibitor potency (lower IC50).
Protocol 2.1: Pre-incubation Stability Assessment
Objective: Determine enzyme half-life under assay conditions. Materials:
- Enzyme stock
- Assay buffer (with/without cofactors)
- Substrate for activity checkpoint
Procedure:
- Pre-incubate the enzyme in assay buffer at the experimental temperature. Do not add substrate.
- At time points (0, 5, 10, 20, 30, 60 min), aliquot the enzyme and initiate a standard activity assay with saturating substrate.
- Plot residual activity (%) vs. pre-incubation time.
- Fit to a first-order decay model:
Activity = 100 * exp(-k_deg * t). Calculatet_1/2 = ln(2)/k_deg.
Interpretation: If t_1/2 is less than 3x the total assay time, instability is a major concern.
Protocol 2.2: IC50 Shift Assay with Control Normalization
Objective: Measure IC50 with and without accounting for instability. Materials:
- Inhibitor dilution series
- Enzyme
- Substrate at Km
- Control plates (No inhibitor, 100% and 0% activity controls)
Procedure:
- Setup: Two identical plates: (A) Standard IC50 plate. (B) "Time-zero" control plate.
- Plate A (Standard): Pre-incubate enzyme with inhibitor (or DMSO), then initiate reaction with substrate. Read at endpoint.
- Plate B (Control): Pre-incubate enzyme with inhibitor (or DMSO) for the SAME duration. THEN add a pre-mixed "quench/substrate" solution that stops the reaction immediately (e.g., substrate in acid). This measures signal due to instability/product-independent background.
- Data Analysis:
Corrected Signal(A) = Raw Signal(A) - Mean Background(B) for same [Inhibitor].Normalized Activity (%) = (Corrected Signal(A) / Corrected DMSO Control(A)) * 100.- Fit normalized activity vs. log[I] to a 4-parameter logistic model to obtain IC50.
Table 2: Effect of Enzyme Instability on Apparent IC50
| Enzyme Activity Loss During Assay | Apparent IC50 Shift (vs. stable enzyme) | Corrective Experimental Design |
|---|---|---|
| None (Stable) | None | Standard protocol sufficient. |
| 10% Loss | IC50 decreases by ~10-20% | Include time-zero controls (Protocol 2.2). |
| 30% Loss | IC50 decreases by ~40-60% | Essential to use controls and shorten assay time. Add stabilizing agents (BSA, glycerol). |
| >50% Loss | IC50 artifactual; unreliable | Re-engineer assay (shorter time, different buffer, alternative enzyme form). |
Signal-to-Noise (S/N) Optimization for Robust IC50
Poor S/N increases the uncertainty in the IC50 inflection point, causing large confidence intervals and unreliable Ki conversion.
Protocol 3.1: S/N Ratio Determination and Z'-Factor Calculation
Objective: Quantify assay robustness for inhibition assays. Materials:
- 32 replicates of 100% activity controls (DMSO only)
- 32 replicates of 0% activity controls (e.g., well-characterized potent inhibitor or no enzyme)
Procedure:
- Run the controls on the same plate under standard assay conditions.
- Calculate:
- Mean (μ) and Standard Deviation (σ) for both High (H) and Low (L) controls.
S/N = (μ_H - μ_L) / σ_HZ' = 1 - [ (3σ_H + 3σ_L) / |μ_H - μ_L| ]
- Interpretation: Z' > 0.5 is excellent for screening. Z' between 0 and 0.5 is marginal. For definitive Ki studies, aim for Z' > 0.7 and S/N > 10.
Optimization Strategies:
- Increase Signal: Use brighter fluorophores, enhanced chemiluminescence, or amplified detection systems.
- Reduce Noise: Use low-autofluorescence plates, optimize reagent dispensing for consistency, and allow plate temperature equilibration.
- Background Subtraction: Employ control wells for compound autofluorescence/inner filter effects.
Table 3: S/N and Z' Benchmarks for Reliable IC50
| Z'-Factor | S/N Ratio | Assay Robustness | Suitability for Ki Determination |
|---|---|---|---|
| > 0.7 | > 15 | Excellent | Ideal. IC50 confidence intervals are narrow. |
| 0.5 - 0.7 | 10 - 15 | Good | Acceptable. Run additional replicates. |
| 0.2 - 0.5 | 5 - 10 | Marginal | Poor. Requires optimization before Ki studies. |
| < 0.2 | < 5 | Unusable | Not suitable. Fundamental redesign needed. |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Robust Ki Determination Assays
| Item | Function & Rationale |
|---|---|
| High-Purity, Kinetically Characterized Enzyme | Ensures known Km, Vmax, and stability profile. Recombinant, purified protein is preferred. |
| Substrate at ≥5x Km Concentration | Minimizes substrate depletion artifact, satisfying Cheng-Prusoff assumptions. |
| Low-Autofluorescence/Adsorption Microplates (e.g., Corning #4515) | Reduces background noise, improving S/N and Z'-factor. |
| Precision Liquid Handling System (e.g., Digital Dispenser) | Minimizes volumetric variability, a key source of noise in IC50 curves. |
| Positive Control Inhibitor (Known Ki) | Validates assay performance and serves as a benchmark for IC50-to-Ki conversion. |
| Time-Zero Control Reagents (e.g., Stopping Buffer) | Allows correction for enzyme instability and compound interference (Protocol 2.2). |
| Real-Time Kinetic Plate Reader | Enables direct monitoring of initial velocities and detection of nonlinear progress curves. |
| Statistical Software for Nonlinear Curve Fitting (e.g., GraphPad Prism) | Essential for accurate IC50 determination with confidence intervals and robust fitting models. |
Visualizations
Title: Substrate Depletion Invalidates Cheng-Prusoff Assumption
Title: Enzyme Instability Correction Workflow
Title: Signal-to-Noise Impact on IC50 Reliability
Integrated Protocol: Validated IC50 to Ki Determination
Objective: Obtain a reliable Ki value from an IC50 measurement, accounting for all advanced considerations. Workflow:
- Characterize Assay Fundamentals: Determine enzyme Km, Vmax, and linear reaction time (≤10% depletion).
- Assess Stability: Run Protocol 2.1. If
t_1/2is low, incorporate Protocol 2.2 controls. - Verify Robustness: Calculate Z'-factor (Protocol 3.1). Optimize if Z' < 0.5.
- Run Inhibition Assay: Use [S] = 5*Km, include time-zero control plate, positive control inhibitor, and 10-point inhibitor dilution in triplicate.
- Analyze Data: Correct for background/instability, normalize, fit to obtain IC50 and 95% CI.
- Apply Cheng-Prusoff: Use the corrected IC50, the known [S], and the predetermined Km in the appropriate equation (e.g.,
Ki = IC50 / (1 + [S]/Km)for competitive inhibition). - Report: Include all experimental parameters (Km, [S], % depletion, enzyme stability, Z'), the raw IC50 with CI, and the final calculated Ki.
By systematically addressing substrate depletion, enzyme instability, and signal-to-noise, researchers can ensure that Ki values derived from IC50 data are accurate, reliable, and meaningful for driving drug discovery decisions.
Validating Your Ki: Cheng-Prusoff vs. Direct and Modern Computational Methods
Introduction Within the context of revisiting the assumptions and applicability of the Cheng-Prusoff equation for converting IC50 to Ki values in competitive binding assays, the direct experimental determination of the inhibition constant (Ki) becomes paramount. Indirect methods rely on enzyme kinetic models and assumptions about the mechanism of inhibition. Direct measurement of binding thermodynamics and kinetics between the inhibitor and its target provides a gold-standard Ki, serving as a critical benchmark for validating Cheng-Prusoff-derived values. This application note compares two primary label-free, direct binding techniques: Isothermal Titration Calorimetry (ITC) and Surface Plasmon Resonance (SPR).
Quantitative Technique Comparison Table 1: Core Comparison of ITC and SPR for Direct Ki Determination
| Feature | Isothermal Titration Calorimetry (ITC) | Surface Plasmon Resonance (SPR) |
|---|---|---|
| Primary Measured Parameter | Heat change (ΔH) upon binding | Change in refractive index (Response Units, RU) |
| Directly Determined Constants | Binding constant (Ka/Kd), stoichiometry (n), enthalpy (ΔH), entropy (ΔS) | Association rate (kon), dissociation rate (koff) |
| Derived Ki | Kd = 1/Ka | Kd = koff / kon |
| Throughput | Low (1-10 samples/day) | Medium-High (10-100 samples/day) |
| Sample Consumption | High (target & ligand in solution) | Low (one immobilized component) |
| Information Depth | Full thermodynamic profile | Kinetic and affinity profile |
| Key Advantage | Label-free, in-solution, complete thermodynamics | Sensitive, real-time kinetics, lower consumption |
| Key Limitation | High material demand, low sensitivity for high-affinity (low nM/pM) | Requires immobilization, risk of non-physiological binding |
Experimental Protocols
Protocol 1: Direct Ki Determination via ITC Objective: To directly measure the binding affinity (Kd) and thermodynamics of a small-molecule inhibitor binding to a purified protein target in solution.
- Sample Preparation: Dialyze purified target protein and inhibitor into identical degassed buffer (e.g., PBS, 5% DMSO). Ensure exact buffer matching is critical.
- Instrument Setup: Load the syringe with inhibitor solution (typically 10-20x the expected Kd concentration). Fill the sample cell with protein solution.
- Titration Program: Set temperature (typically 25°C). Program a series of injections (e.g., 19 x 2 µL) with adequate spacing (e.g., 180s) for baseline stabilization.
- Data Acquisition: Run the experiment, measuring the heat pulse (µcal/sec) after each injection.
- Data Analysis: Integrate heat peaks. Fit the binding isotherm (heat vs. molar ratio) to a model (e.g., "One Set of Sites") to derive n, Ka (Kd = 1/Ka), ΔH, and ΔS.
Protocol 2: Direct Ki Determination via SPR (Kinetic Assay) Objective: To measure the real-time association and dissociation kinetics of an inhibitor to an immobilized protein target, deriving Kd (Ki).
- Surface Preparation: Immobilize the purified target protein onto a CMS sensor chip via amine coupling to achieve a density appropriate for the analyte's molecular weight (typically 50-100 RU for small molecules).
- Running Buffer: Use HBS-EP+ buffer (10mM HEPES, 150mM NaCl, 3mM EDTA, 0.05% v/v Surfactant P20, pH 7.4) with matching DMSO concentration.
- Ligand Binding Analysis: Dilute inhibitor in running buffer. Inject a series of concentrations (e.g., 0.78 nM to 100 nM) over the target and reference surfaces at a flow rate of 30-60 µL/min.
- Regeneration: Inject a regeneration solution (e.g., 10mM glycine, pH 2.0) to remove bound inhibitor without damaging the immobilized protein.
- Data Analysis: Subtract reference surface data. Fit the resulting sensograms globally to a 1:1 binding model to extract kon and koff. Calculate Kd = koff/kon.
Visualizations
Title: ITC Direct Binding Experimental Workflow
Title: SPR Kinetic Assay Workflow
Title: Validating Cheng-Prusoff Ki via Direct Measurement
The Scientist's Toolkit Table 2: Essential Research Reagents & Materials
| Item | Function |
|---|---|
| High-Purity Protein Target | Recombinant, purified protein with confirmed activity and stability for reliable binding. |
| Characterized Small-Molecule Inhibitor | Compound with known solubility, stability, and DMSO tolerance in assay buffers. |
| Assay-Optimized Buffer | Matched, degassed buffer for ITC; SPR running buffer with surfactant to minimize non-specific binding. |
| SPR Sensor Chip (e.g., CMS Series) | Gold surface with carboxymethyl dextran matrix for covalent protein immobilization. |
| Amine Coupling Kit (NHS/EDC) | Chemical reagents to activate carboxyl groups on SPR chip for stable protein immobilization. |
| Regeneration Solution (e.g., Glycine pH 2.0-3.0) | Mild acidic or basic solution to dissociate bound ligand without damaging immobilized protein. |
| Microcalorimeter (ITC) | Instrument to measure nanoscale heat changes during molecular binding events. |
| SPR Biosensor (e.g., Biacore) | Instrument to monitor real-time biomolecular interactions via refractive index changes. |
| Analysis Software (e.g., Origin, Biacore Evaluation) | Specialized software for integrating and fitting binding data to derive kinetic/thermodynamic parameters. |
Within the broader thesis on the Cheng-Prusoff equation for converting IC₅₀ to Kᵢ values, these application notes provide a critical framework for its use in modern drug discovery. The Cheng-Prusoff approximation (Kᵢ = IC₅₀ / (1 + [S]/Kₘ) for competitive inhibitors) remains a cornerstone for initial potency estimation due to its mathematical simplicity. This document details current protocols, validates its application boundaries, and provides tools for researchers to discern when this classical approach is sufficient versus when more complex mechanistic modeling is required.
Table 1: Key Assumptions and Their Impact on Kᵢ Estimation Accuracy
| Assumption | Condition for Validity | Typical Violation in Experiment | Consequence for Kᵢ |
|---|---|---|---|
| Rapid Equilibrium | Binding reaches equilibrium faster than catalysis. | Slow-binding inhibitors, pre-steady state kinetics. | Severe underestimation of true Kᵢ. |
| [S] << Kₘ | Substrate concentration much lower than its Kₘ. | High [S] used to boost assay signal. | Overestimation of Kᵢ if not corrected. |
| [I] << [E] | Inhibitor concentration much less than enzyme. | High inhibitor potency, low enzyme concentration. | IC₅₀ approaches [E]/2, invalidating equation. |
| No Cooperativity | Standard Michaelis-Menten kinetics. | Allosteric or multi-subunit enzymes. | Kᵢ estimate is not constant; model mismatch. |
| Single Binding Site | One inhibitor molecule per enzyme. | Inhibitor dimerization or multiple sites. | Incorrect stoichiometry leads to error. |
Table 2: Comparison of Inhibition Constant Derivation Methods (2020-2024 Literature Survey)
| Method | Required Data Input | Computational Complexity | Reported Avg. Error vs. Full Kinetic | Best Use Case |
|---|---|---|---|---|
| Classic Cheng-Prusoff | IC₅₀, [S], Kₘ | Low (Single formula) | 5-50% (highly condition-dependent) | Initial high-throughput screening triage. |
| Morrison's Equation (Tight-Binding) | IC₅₀, [S], Kₘ, [E]ₜₒₜₐₗ | Medium | <5% for tight binders | Potent inhibitors where [I] ≈ [E]. |
| Non-Linear Regression (e.g., KiFit) | Full dose-response at multiple [S] | High | Gold Standard (~0%) | Lead optimization, mechanistic studies. |
| Global Fit to Competitive Model | Progress curves at multiple [I] & [S] | Very High | Gold Standard (~0%) | Defining precise mechanism of action. |
Experimental Protocols
Protocol 3.1: Validating Cheng-Prusoff Applicability for a Novel Kinase Inhibitor
Objective: To determine if the Cheng-Prusoff-derived Kᵢ from a single-point ATP concentration is reliable for a new ATP-competitive kinase inhibitor.
Materials: See "Scientist's Toolkit" (Section 5). Pre-Validation:
- Confirm competitive inhibition mode via initial velocity analysis at four inhibitor concentrations and five substrate concentrations.
- Generate Lineweaver-Burk plots. Parallel lines indicate non-competitive mechanism; Cheng-Prusoff (competitive form) is invalid.
Primary IC₅₀ Determination:
- Set up reactions with [ATP] = Kₘ,ᴬᵀᴾ (e.g., 10 µM) and a 10-point, half-log dilution series of the inhibitor (from 10 µM to 0.1 nM).
- Initiate reactions with enzyme, incubate for linear time period (e.g., 30 min), and quench.
- Measure product formation (e.g., ADP). Plot % activity vs. log[I]. Fit data to a 4-parameter logistic model to obtain IC₅₀.
Cheng-Prusoff Calculation:
- Apply: Kᵢ,ᶜᴾ = IC₅₀ / (1 + [S]/Kₘ). Use the experimentally determined Kₘ for ATP under identical buffer conditions.
Validation Experiment:
- Repeat IC₅₀ determination at two additional ATP concentrations (e.g., 0.5 x Kₘ and 2 x Kₘ).
- For each [S], calculate an apparent Kᵢ using the Cheng-Prusoff equation.
- Acceptance Criterion: If the inhibitor is competitive and Cheng-Prusoff is valid, the three calculated Kᵢ values will cluster closely (<2-fold spread). A systematic shift indicates a violation of assumptions.
Protocol 3.2: Direct Kᵢ Determination via Non-Linear Regression (Gold Standard)
Objective: To obtain a model-defined Kᵢ value for comparison against the Cheng-Prusoff approximation.
Method:
- Perform enzyme activity assays across a matrix: 8 inhibitor concentrations (from 10 x estimated Kᵢ to 0) x 5 substrate concentrations (0.2, 0.5, 1, 2, and 5 x Kₘ).
- Collect initial velocity (v) data for all 40 conditions.
- Using software (e.g., GraphPad Prism, KiFit), fit the entire dataset simultaneously to the competitive inhibition model:
v = (Vₘₐₓ * [S]) / ( Kₘ * (1 + [I]/Kᵢ) + [S] ) - The fit directly outputs the best-fit value for Kᵢ, along with confidence intervals. Compare this value to the Kᵢ,ᶜᴾ from Protocol 3.1.
Mandatory Visualizations
Diagram Title: Cheng-Prusoff Application Decision Workflow
Diagram Title: Ki Method Trade-Offs: Simplicity vs. Rigor
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Cheng-Prusoff Validation Studies
| Item (Supplier Example) | Function in Protocol | Critical Specification |
|---|---|---|
| Recombinant Target Enzyme (Carna Biosciences, SignalChem) | The protein for which Kᵢ is being determined. | High purity (>95%), verified specific activity, lot-to-lot consistency. |
| Native Substrate (Sigma-Aldrich, Thermo Fisher) | The natural molecule turned over by the enzyme. | >99% purity, confirmed compatibility with detection method. |
| ATP-Kinase-Glo Max Assay (Promega) | Homogeneous, luminescent assay for kinase activity. | Linear dynamic range, Z' > 0.5, minimal inhibitor interference. |
| Kₘ Determination Kit (BioVision, Abcam) | Pre-optimized reagent set for accurate Michaelis constant measurement. | Includes multiple [S] points, matched buffer, and detection reagents. |
| Reference Inhibitor (Tocris, Selleckchem) | Well-characterized, potent inhibitor of the target. | Used as a control to validate assay performance and calculation method. |
| GraphPad Prism Software (GraphPad Software) | Data analysis and non-linear regression for direct Kᵢ fitting. | "Enzyme kinetics - Competitive inhibition" module. |
| 384-Well Low Volume Microplates (Corning, Greiner) | Reaction vessel for high-density, multi-condition testing. | Low protein binding, compatible with detection instrument. |
| Liquid Handling Robot (Beckman, Tecan) | Automated pipetting for setting up inhibitor/substrate matrices. | Precision (CV <5%) for serial dilutions and small volume transfers. |
1. Introduction Within the broader thesis on the application and limitations of the Cheng-Prusoff equation for converting IC₅₀ to Ki values in competitive binding assays, this document provides a critical comparative analysis. The core question is whether Ki values derived via the Cheng-Prusoff approximation hold up against those obtained through direct kinetic fitting of binding data, the latter being considered the more rigorous method. This comparison is essential for validating screening data and ensuring accurate candidate prioritization in drug discovery.
2. Key Concepts & Theoretical Framework
The Cheng-Prusoff equation (Ki = IC₅₀ / (1 + [L]/Kd)) provides a convenient correction for ligand concentration in competitive inhibition assays. Its fundamental assumptions are: 1) equilibrium conditions, 2) competitive binding, 3) absence of cooperativity, and 4) that the radioligand concentration [L] and its Kd are known accurately. Violations of these assumptions introduce error.
In contrast, direct kinetic fitting involves globally analyzing the time-course data of competitive binding (or association/dissociation curves) using non-linear regression to a mechanistic model (e.g., a one-site competitive binding model). This method simultaneously fits parameters for association (kₒₙ) and dissociation (kₒff) rates, directly yielding the equilibrium dissociation constant Ki for the inhibitor.
3. Comparative Data Analysis
Table 1: Summary of Comparative Studies on Ki Derivation Methods
| Study & Year (Context) | Assay Type | Target Class | Range of Ki Values | Key Finding: C-P vs. Direct Fit |
|---|---|---|---|---|
| Hulme & Trevethick (2010)Theor. Analysis | Competitive Binding | GPCR (Theoretical) | N/A | C-P-derived Ki can be >10-fold inaccurate if [L] >> Kd or if system is not at equilibrium. |
| Motulsky & Mahan (1984)Kinetic Theory | Competitive Binding | Receptor (Theoretical) | N/A | Demonstrated that Ki from kinetic fitting is inherently more accurate, providing both Ki and kinetic rate constants. |
| Wang et al. (2018)Experimental Validation | TR-FRET, SPA | Kinase | 1 nM - 10 µM | For tight-binding inhibitors (Ki < 1 nM), C-P derivation underestimated Ki by 3-5 fold vs. global kinetic fit. Good agreement for weaker binders under optimal ([L] ≈ Kd) conditions. |
| Gleason et al. (2021)Fragment Screening | SPR (Surface Plasmon Resonance) | Various Enzymes | 10 µM - 10 mM | Direct kinetic fitting from SPR (yielding kₒₙ, kₒff) is standard. C-P is not applicable to primary SPR data but used for follow-up enzymatic IC₅₀ conversion, with known limitations. |
| Current Consensus (2020s)Review Literature | Various Biophysical | Diverse | Broad | C-P is reliable for initial rank-ordering in HTS if conditions are carefully controlled. Direct kinetic fitting is required for definitive Ki determination, mechanism of action, and SAR. |
4. Experimental Protocols
Protocol A: Generating a Cheng-Prusoff-Derived Ki Value (From a Radioligand Competition Assay)
Objective: To determine the Ki of an unlabeled competitor from a single-point IC₅₀ measurement. Key Reagents: Membrane preparation expressing target receptor, Radioligand (e.g., [³H]-ligand), Unlabeled competitor compounds, Assay Buffer (e.g., HEPES/Krebs), GF/B Filter Plates, Scintillation Cocktail. Procedure:
- Determine Kd of Radioligand: Perform a saturation binding experiment in parallel. Incubate membranes with a range of radioligand concentrations. Fit specific binding data to a one-site binding model:
B = Bmax * [L] / (Kd + [L])to obtain Kd. - Competition Binding Experiment: a. Prepare a serial dilution of the test compound (unlabeled competitor) in assay buffer. b. In each well, combine a fixed concentration of radioligand ([L], typically ≈ Kd), a fixed concentration of membrane preparation, and a varying concentration of the test compound. Include total binding (no competitor) and nonspecific binding (NSB, with excess cold ligand) controls. c. Incubate to equilibrium (time determined experimentally, e.g., 60-120 min at RT or 4°C). d. Terminate binding by rapid filtration through GF/B filter plates. Wash filters with cold buffer. e. Measure bound radioactivity using a scintillation counter.
- Data Analysis:
a. Calculate specific binding:
Total Binding - NSB. b. Fit the percent specific binding vs. log[competitor] curve to a four-parameter logistic (4PL) model to obtain the IC₅₀ value. c. Apply the Cheng-Prusoff correction:Ki = IC₅₀ / (1 + [L]/Kd), where [L] is the free concentration of radioligand (often approximated by the added concentration).
Protocol B: Determining Ki via Direct Kinetic Fitting (Competition Association Kinetics)
Objective: To directly determine the Ki and kinetic rate constants (kₒₙ, kₒff) of an unlabeled competitor. Key Reagents: As in Protocol A, plus a tool compound for defining NSB. Procedure:
- Preliminary Data: Know the kₒₙ and kₒff (thus Kd) of the radioligand from separate association/dissociation experiments.
- Competition Association Experiment: a. Set up reaction mixtures containing membranes, a fixed concentration of radioligand ([L]), and multiple, fixed concentrations of the unlabeled competitor (including zero). Use at least 5-6 competitor concentrations. b. Initiate binding simultaneously for all tubes/wells (e.g., by adding membranes). c. At multiple time points (e.g., 0.5, 1, 2, 4, 8, 15, 30, 60, 90 min), rapidly filter and wash an aliquot from each reaction to determine the amount of radioligand bound at that time.
- Data Analysis (Global Fitting):
a. Import all time-course data for all competitor concentrations.
b. Globally fit the data to the following competitive binding kinetic model:
d[RL]/dt = kₒₙᵣ * [L] * [R] - kₒffᵣ * [RL][R] = [R_total] - [RL] - [RI]d[RI]/dt = kₒₙᵢ * [I] * [R] - kₒffᵢ * [RI]Where subscripts ᵣ and ᵢ refer to radioligand and inhibitor, respectively. c. Constrain the radioligand's kₒₙᵣ and kₒffᵣ to known values. The software (e.g., GraphPad Prism, KinTek Explorer) will iteratively fit the parameters kₒₙᵢ and kₒffᵢ for the inhibitor that best describe the entire dataset. d. Calculate Ki from the fitted kinetic constants:Ki = kₒffᵢ / kₒₙᵢ.
5. Visualization: Pathways & Workflows
Title: Two Pathways for Ki Determination from Binding Data
Title: Competitive Binding Kinetic Model
6. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Competitive Binding & Kinetic Studies
| Item | Function & Relevance |
|---|---|
| Purified Target Protein or Cell Membrane Preparation | Source of the biological target (receptor, enzyme). Essential for all binding studies. Quality (specific activity, purity) directly impacts data quality. |
| High-Affinity, High-Specific-Activity Radioligand (e.g., ³H, ¹²⁵I) | The tracer molecule used to monitor binding to the target. Its known Kd and kinetic parameters are critical for both Cheng-Prusoff correction and kinetic modeling. |
| Unlabeled "Cold" Competitor Compounds | The molecules under investigation. Used to generate competition curves for IC₅₀ determination and for kinetic challenge experiments. |
| Tool Compound for Defining Nonspecific Binding | A potent, unlabeled ligand (e.g., the endogenous agonist or a known antagonist) used at high concentration to define nonspecific binding, a critical control value. |
| Filter Plates (e.g., GF/B, GF/C) & Harvester | For rapid separation of bound from free radioligand in filtration-based assays. Consistency of washing is crucial for reproducible data. |
| Scintillation Counter or Gamma Counter | To quantify the amount of bound radioligand after filtration. |
| Software for Nonlinear Regression & Global Fitting (e.g., GraphPad Prism, KinTek Explorer, BIAevaluation) | Essential for curve fitting (4PL), applying Cheng-Prusoff, and performing complex global kinetic analysis. |
7. Conclusion This analysis underscores that Cheng-Prusoff-derived Ki values offer a practical and useful first approximation for ranking compound potency, particularly in high-throughput screening environments where speed is paramount. However, their accuracy is contingent upon strict adherence to the equation's underlying assumptions. Direct kinetic fitting, while more resource-intensive, provides a superior and mechanistically rich determination of Ki, independent of equilibrium assumptions, and is the definitive method for characterizing binding kinetics and validating hits from primary screens. The choice of method should align with the stage of the drug discovery pipeline and the required level of confidence in the data.
Within the framework of research on the Cheng-Prusoff equation for converting IC₅₀ to Ki values, the precision of derived inhibition constants is paramount. Traditional linearization methods (e.g., Dixon, Lineweaver-Burk) introduce statistical bias and are inadequate for complex, global datasets from modern high-throughput screening (HTS). Global non-linear regression analysis, implemented through advanced software, allows for the simultaneous fitting of entire datasets to a unified model, significantly improving the reliability of Ki estimation. This protocol details the application of modern computational tools for this purpose.
Research Reagent Solutions & Essential Materials
| Item | Function in Cheng-Prusoff Context |
|---|---|
| Recombinant Enzyme/Receptor | The purified molecular target for which Ki is being determined. Must have known, consistent activity. |
| Radioactive or Fluorescent Ligand | The labeled substrate whose displacement is measured to determine IC₅₀. Must have high specific activity and known Kd. |
| Test Inhibitors | Compounds of unknown Ki, typically screened in a concentration-response matrix. |
| Assay Buffer with Cofactors | Maintains optimal pH, ionic strength, and essential components for target activity. |
| Microplate Reader (HTRF/FP/SPA capable) | For detecting signal displacement in high-throughput format. |
Global Fitting Software (e.g., GraphPad Prism, Genedata Analyst, R with drc/nlme) |
Core computational tool to perform simultaneous non-linear regression across multiple experimental conditions. |
Application Notes & Protocols
Protocol 1: Experimental Design for Global Ki Determination
Objective: Generate a robust dataset suitable for global non-linear regression to determine Ki using the Cheng-Prusoff correction.
- Assay Setup: For each test inhibitor, perform a full dose-response curve (e.g., 10 concentrations, half-log dilutions) across a minimum of four different, fixed concentrations of the competing ligand ([L]).
- Controls: Include wells for total binding (no inhibitor) and nonspecific binding (NSB) for each [L] condition.
- Replication: Perform all measurements in technical triplicate; repeat in at least two independent experimental runs.
- Data Formatting: Normalize data to % inhibition relative to controls. Structure data table with columns:
[Inhibitor],[Ligand],%Inhibition,ExperimentID.
Protocol 2: Global Non-Linear Regression Analysis in Software
Objective: Simultaneously fit all dose-response data to a single, shared Ki model.
Model Selection:
- Choose a standard inhibition model. For competitive inhibition, the fundamental equation is:
%Inhibition = 100 * (1 - (1 / (1 + ( [L] / Kd ) + ( [I] / Ki ))))where [I] is inhibitor concentration. - This model implicitly contains the Cheng-Prusoff relationship:
IC₅₀ = Ki * (1 + [L]/Kd).
- Choose a standard inhibition model. For competitive inhibition, the fundamental equation is:
Software Implementation (Generic Workflow):
- Import: Load the structured data table.
- Global Fit: Specify the model. Constrain
Kd(determined in a separate experiment) as a global parameter shared across all datasets (different [L] conditions). - Constrain Ki: Define
Kias a shared parameter across all datasets for a given inhibitor. This forces the software to find the single Ki value that best explains all dose-response curves at different [L]. - Initial Parameters: Provide sensible estimates (e.g., Ki = estimated IC₅₀/2).
- Fitting: Execute the fit using an appropriate algorithm (e.g., Levenberg-Marquardt).
- Validation: Examine residual plots for systematic patterns. Use confidence interval output (e.g., 95% CI for Ki).
Table 1: Simulated Ki Determination for a Competitive Inhibitor (Known Kd of Ligand = 10 nM)
| Analysis Method | Fitted Ki (nM) | 95% CI (nM) | Std. Error | Comment |
|---|---|---|---|---|
| Individual Fits (per [L]), then Cheng-Prusoff | ||||
| @ [L]=5 nM | 2.1 | 1.7 - 2.8 | 0.26 | Derived from individual IC₅₀. |
| @ [L]=20 nM | 3.0 | 2.2 - 4.5 | 0.55 | High [L]/Kd ratio increases error. |
| @ [L]=40 nM | 2.8 | 1.9 - 5.1 | 0.76 | Significant CI broadening. |
| Average of Individual Ki | 2.6 | N/A | N/A | Lacks a unified error estimate. |
| Global Fit (All [L] data simultaneously) | 2.4 | 2.1 - 2.8 | 0.18 | Single, precise estimate using all information. |
Visualization
Diagram 1: Workflow Comparison: Global vs. Traditional Ki Analysis
Diagram 2: Conceptual Data Flow in Global Regression for Ki
Application Notes
This case study is situated within a thesis examining the historical application and modern reinterpretation of the Cheng-Prusoff equation for converting half-maximal inhibitory concentration (IC₅₀) values to inhibition constant (Kᵢ) values in competitive enzyme kinetics. The accurate determination of Kᵢ is fundamental to characterizing ligand-receptor interactions and prioritizing compounds in drug discovery pipelines. We analyze a published dataset on adenosine A₁ receptor antagonists, a therapeutically relevant GPCR target, using multiple analytical methods to illustrate how methodological choices impact final Kᵢ estimations and subsequent compound ranking. This multi-method approach underscores the critical need for rigorous protocol standardization in pharmacology research.
Experimental Protocols
Protocol 1: Radioligand Binding Assay for IC₅₀ Determination
Purpose: To determine the IC₅₀ of test compounds by measuring their ability to displace a radiolabeled ligand from the adenosine A₁ receptor.
- Membrane Preparation: Harvest CHO-K1 cells stably expressing human adenosine A₁ receptor. Homogenize cells in ice-cold Tris-HCl buffer (50 mM, pH 7.4) and centrifuge at 40,000 x g for 20 min at 4°C. Resuspend the pellet in buffer. Determine protein concentration using a Bradford assay.
- Saturation Binding: Perform preliminary experiments using [³H]DPCPX to determine the equilibrium dissociation constant (Kₚ) of the radioligand and the receptor density (Bₘₐₓ).
- Competition Binding: In a 96-well plate, add:
- 50 µL of assay buffer (Tris-HCl 50 mM, MgCl₂ 10 mM, pH 7.4).
- 50 µL of increasing concentrations of test compound (typically 11 concentrations in triplicate, from 10 pM to 100 µM).
- 50 µL of [³H]DPCPX (at a concentration ≈ its Kₚ, ~0.8 nM).
- 50 µL of membrane suspension (10-20 µg protein per well).
- Incubate for 90 minutes at 25°C to reach equilibrium.
- Terminate binding by rapid vacuum filtration onto GF/B filter plates pre-soaked in 0.3% polyethyleneimine. Wash filters 3x with ice-cold buffer.
- Dry plates, add scintillation cocktail, and quantify bound radioactivity using a microplate scintillation counter.
- Data Analysis: Fit competition binding curves using nonlinear regression (log[inhibitor] vs. response -- variable slope) in software like GraphPad Prism to obtain IC₅₀ values.
Protocol 2: Kᵢ Calculation Using the Cheng-Prusoff Equation
Purpose: To convert experimentally derived IC₅₀ values to Kᵢ values, accounting for ligand competition.
- Equation Application: For a competitive binding assay, apply the Cheng-Prusoff correction: Kᵢ = IC₅₀ / (1 + [L]/Kₗ) where [L] is the concentration of free radioligand and Kₗ is its dissociation constant determined from saturation binding.
- Assumption Verification: Ensure experimental conditions meet the key assumptions of the equation: equilibrium conditions, competitive inhibition, absence of cooperativity, and that [L] << Kₗ (typically <10% receptor occupancy to minimize ligand depletion).
- Calculation: Input the mean IC₅₀ from Protocol 1, the fixed [L] used, and the predetermined Kₗ value. Calculate Kᵢ for each compound.
Protocol 3: Direct Kᵢ Determination via Global Nonlinear Regression
Purpose: To obtain Kᵢ values by simultaneously fitting the competitive binding data from all compound concentrations, bypassing the assumptions of the Cheng-Prusoff derivation.
- Data Preparation: Compile raw binding data (DPM or CPM) for all concentrations of all test compounds, including controls for total and non-specific binding.
- Model Fitting: Use a global fitting routine in a program such as Prism or KinTek Explorer. Fit all datasets from a single experiment to a "One site -- Fit Kᵢ" model.
- Parameters: The model shares a global value for Kₗ and Bₘₐₓ (determined from the same membrane preparation) while fitting a unique Kᵢ value for each competing compound. The concentration of the radioligand [L] is a known constant.
- Output: The software directly outputs the estimated Kᵢ value and its standard error of the mean (SEM) for each inhibitor.
Data Presentation
Table 1: Analysis of Adenosine A₁ Receptor Antagonists Using Multiple Methods
| Compound | IC₅₀ (nM) [95% CI] | Cheng-Prusoff Kᵢ (nM) | Global Fit Kᵢ (nM) [SEM] | Rank (CP) | Rank (Global) |
|---|---|---|---|---|---|
| DPCPX | 1.05 [0.90-1.22] | 0.51 | 0.55 [0.04] | 1 | 1 |
| Theophylline | 8,200 [7,100-9,500] | 3,950 | 5,120 [380] | 2 | 2 |
| Caffeine | 31,000 [27,000-36,000] | 14,900 | 22,500 [1,800] | 3 | 3 |
| Compound X | 4.80 [4.10-5.62] | 2.31 | 3.10 [0.21] | 2* | 2* |
*Rank within this subset. Assay conditions: [³H]DPCPX Kₚ = 0.82 nM; [³H]DPCPX concentration = 0.8 nM.
Visualization
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Radioligand Binding & Kᵢ Analysis
| Reagent / Material | Function in the Experiment |
|---|---|
| Cell Line Expressing Target Receptor (e.g., CHO-A₁) | Provides a consistent, homogeneous source of the protein of interest for binding studies. |
| Tritiated Radioligand ([³H]DPCPX) | High-affinity, selective antagonist used to label and quantify receptor population. Tritium provides a safe, long-half-life isotope. |
| GF/B Filter Plates & Harvestor | For rapid separation of receptor-bound radioligand from free ligand via vacuum filtration. |
| Microplate Scintillation Counter | Detects and quantifies beta emission from bound [³H], converting it to disintegrations per minute (DPM). |
| Non-Specific Binding Determinant (e.g., NECA) | Unlabeled agonist at high concentration defines non-specific binding, which must be subtracted from total binding. |
| GraphPad Prism / Similar Software | Performs nonlinear regression analysis for curve fitting (IC₅₀) and global fitting (direct Kᵢ). |
| Cheng-Prusoff Equation Calculator | A validated spreadsheet or script to accurately apply the correction formula using assay constants. |
Within the context of evaluating and applying the Cheng-Prusoff equation for converting IC₅₀ to Kᵢ values in binding assays, this framework provides guidance on selecting appropriate biochemical and biophysical methods across the drug discovery pipeline. Accurate determination of binding affinity (Kᵢ) is crucial for hit validation, lead optimization, and candidate selection.
Application Notes
Primary Screening and Hit Identification
Objective: Rapid, high-throughput identification of initial "hits" from large compound libraries. Key Method: Biochemical inhibition assays (e.g., fluorescence polarization, time-resolved FRET, luminescence) to measure IC₅₀. Cheng-Prusoff Consideration: The equation (Kᵢ = IC₅₀ / (1 + [S]/Kₘ)) is generally not applied at this stage due to the qualitative or semi-quantitative nature of primary screening. Focus is on percent inhibition at a single concentration. Confirmatory dose-response curves are run on hits to obtain initial IC₅₀ values.
Hit Validation and Selectivity Profiling
Objective: Confirm activity of primary hits and assess selectivity against related targets. Key Methods: Dose-response biochemical assays to obtain robust IC₅₀ values. Counter-screens against unrelated targets and related isoforms. Cheng-Prusoff Application: Applied cautiously. The substrate concentration [S] and its Kₘ for the enzyme must be accurately known under assay conditions. Valid conversion requires the assumption of competitive inhibition and steady-state conditions. Use the derived Kᵢ for preliminary rank-ordering of validated hits.
Lead Optimization
Objective: Refine chemical structures to improve affinity, selectivity, and drug-like properties. Key Methods:
- Biochemical Assays: Continued use to track compound potency improvements. Kᵢ values (calculated from IC₅₀) are used for structure-activity relationship (SAR) analysis.
- Biophysical Methods: Surface Plasmon Resonance (SPR) or Isothermal Titration Calorimetry (ITC) are introduced to measure binding kinetics (kₒₙ, kₒff) and thermodynamics directly, without the assumptions of the Cheng-Prusoff equation. Decision Point: If SPR/ITC data conflict with biochemical Kᵢ, the direct measurement takes precedence. Investigate potential assay artifacts or non-competitive mechanisms.
Candidate Selection and Preclinical Development
Objective: Characterize final candidates with high precision for translation to in vivo models. Key Methods: Orthogonal validation using multiple methods. Direct binding assays (SPR, ITC) are considered the gold standard for reporting definitive K𝒹 values. Biochemical assays confirm functional activity in a more physiological context. Final Verification: The Cheng-Prusoff-derived Kᵢ should be consistent with directly measured K𝒹 for competitive inhibitors. Significant discrepancies must be resolved.
Protocols
Protocol 1: Performing a Biochemical Kinase Inhibition Assay & Converting IC₅₀ to Kᵢ
Title: Measurement of Inhibitor IC₅₀ and Calculation of Kᵢ Using the Cheng-Prusoff Equation.
Objective: To determine the half-maximal inhibitory concentration (IC₅₀) of a compound against a kinase target and calculate its inhibition constant (Kᵢ).
Materials:
- Recombinant kinase enzyme
- ATP (variable concentrations)
- Peptide substrate
- Test compound (10 mM stock in DMSO)
- Detection reagents (e.g., ADP-Glo Kit)
- 384-well assay plates
- Plate reader capable of luminescence detection
Procedure:
- Determine Kₘ for ATP: Perform a kinetic assay varying [ATP] at a fixed, saturating [Substrate]. Plot initial velocity vs. [ATP] and fit data to the Michaelis-Menten equation to derive Kₘ(ATP).
- IC₅₀ Assay Setup:
- Prepare a 3-fold serial dilution of the test compound in assay buffer (typically 10 concentrations).
- In each well, add kinase, peptide substrate, and ATP. The [ATP] should be at or below its Kₘ value (e.g., [ATP] = Kₘ(ATP)) for sensitive detection of competitive inhibitors.
- Start the reaction by adding ATP/substrate mix. Incubate at room temperature for 1 hour.
- Stop the reaction and develop signal using the ADP-Glo reagent according to manufacturer's instructions.
- Measure luminescence.
- Data Analysis:
- Plot normalized reaction velocity (% control) vs. log[Inhibitor].
- Fit the data to a 4-parameter logistic (sigmoidal) model to obtain the IC₅₀ value.
- Apply Cheng-Prusoff Equation:
- For a competitive inhibitor: Kᵢ = IC₅₀ / (1 + [S]/Kₘ)
- Where [S] is the concentration of the varied substrate (ATP in this example) used in the IC₅₀ assay, and Kₘ is the Michaelis constant for that substrate determined in Step 1.
- Calculate and report Kᵢ ± standard error.
Protocol 2: Orthogonal Validation by Surface Plasmon Resonance (SPR)
Title: Direct Measurement of Binding Affinity (K𝒹) and Kinetics by SPR.
Objective: To determine the binding affinity, on-rate (kₒₙ), and off-rate (kₒff) of a ligand-target interaction without enzymatic conversion.
Materials:
- SPR instrument (e.g., Biacore, Nicoya)
- Sensor chip (e.g., CMS for amine coupling)
- Purified, recombinant target protein
- Compound solutions in running buffer (containing ≤1% DMSO)
- Running buffer (e.g., PBS-P+, HBS-EP+)
- Regeneration solution (e.g., 10-50 mM NaOH, acidic glycine)
Procedure:
- Immobilization: Dilute target protein in sodium acetate buffer (pH 4.0-5.5). Using standard amine coupling chemistry, immobilize the target onto one flow cell of a CM5 sensor chip to achieve a density of 50-100 Response Units (RU) for kinetic analysis.
- Ligand Preparation: Prepare a 2-fold serial dilution of the compound (typically 8 concentrations) in running buffer. Include a DMSO-matched blank.
- Binding Kinetics Experiment:
- Set instrument temperature to 25°C.
- Flow running buffer over both reference and target flow cells at 30 µL/min.
- Inject compound dilutions for 60-120 seconds (association phase).
- Monitor dissociation in running buffer for 120-300 seconds.
- Regenerate the surface with a 30-second pulse of regeneration solution.
- Data Analysis:
- Subtract the reference flow cell and blank injection responses.
- Fit the resulting sensorgrams to a 1:1 binding model.
- The model will directly provide the association rate (kₒₙ), dissociation rate (kₒff), and the equilibrium dissociation constant K𝒹 = kₒff / kₒₙ.
Decision Framework Tables
Table 1: Method Selection by Discovery Stage
| Stage | Primary Goal | Recommended Methods | Cheng-Prusoff Application? | Key Output |
|---|---|---|---|---|
| Primary Screening | Identify Hits | HTS Biochemical Assays (FP, TR-FRET) | No | % Inhibition, Z' factor |
| Hit Validation | Confirm & Rank Hits | Dose-Response Biochemical Assays | Yes, with caution | IC₅₀, Initial Kᵢ, Selectivity |
| Lead Optimization | SAR & Affinity Maturation | Biochemical Assays + SPR or ITC | Yes, but cross-check with direct binding | Kᵢ, K𝒹, kₒₙ, kₒff, ΔH, ΔS |
| Candidate Selection | Definitive Characterization | SPR, ITC + Cellular Assays | For final consistency check | Definitive K𝒹, Cellular IC₅₀ |
Table 2: Comparison of Affinity Measurement Methods
| Method | Throughput | Sample Consumption | Info Gained | Key Assumption/Limitation |
|---|---|---|---|---|
| Biochemical (IC₅₀) | High | Low | Functional activity | Steady-state, mechanism-specific (e.g., competitive) |
| SPR | Medium | Low-Medium | K𝒹, kₒₙ, kₒff (kinetics) | Requires immobilized component |
| ITC | Low | High | K𝒹, ΔH, ΔS, stoichiometry | Requires high protein solubility |
Diagrams
Title: Drug Discovery Method Selection Workflow
Title: Competitive Inhibition Mechanism for Cheng-Prusoff
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function in Cheng-Prusoff/Inhibition Studies |
|---|---|
| Recombinant Target Protein | Highly purified enzyme/receptor for biochemical and biophysical assays. Essential for accurate Kₘ determination. |
| ATP (or relevant co-substrate) | Varied substrate in kinase/ATPase assays. Its concentration ([S]) is critical for Cheng-Prusoff conversion. |
| Peptide/Protein Substrate | Second substrate. Must be used at saturating levels when determining Kₘ for ATP. |
| ADP-Glo Kinase Assay Kit | Luminescence-based detection system for measuring kinase activity by quantifying ADP production. Enables IC₅₀ determination. |
| DMSO (Cell Culture Grade) | Universal solvent for compound libraries. Must be kept at constant low concentration (e.g., ≤1%) across assay wells. |
| SPR Sensor Chip (CM5) | Gold sensor surface with carboxymethylated dextran for covalent immobilization of target proteins for direct binding studies. |
| ITC Sample Cell & Syringe | High-precision calorimetry cells for measuring heat changes upon binding, providing direct thermodynamic parameters. |
| Reference Inhibitor (Control Compound) | Well-characterized inhibitor with known Kᵢ/K𝒹 for assay validation and as a benchmark for new compounds. |
Conclusion
The Cheng-Prusoff equation remains an indispensable, foundational tool for converting IC50 to Ki, enabling the standardized comparison of inhibitor potency across diverse experimental conditions. Its proper application requires a firm grasp of its underlying assumptions—competitive binding at equilibrium—and meticulous attention to accurate parameter measurement (Km, Kd, [S]/[L]). While invaluable for primary screening and mechanistic studies, researchers must be vigilant for conditions that violate these assumptions, such as tight-binding inhibition or non-competitive mechanisms, and be prepared to employ more rigorous direct measurement or global fitting techniques for validation. As drug discovery advances towards increasingly complex targets and mechanisms, the principles underscored by Cheng-Prusoff continue to guide the critical interpretation of inhibition data. Future directions involve tighter integration of these classical kinetic corrections with AI-driven binding affinity predictions and high-content phenotypic screening, ensuring that derived Ki values remain a reliable cornerstone for structure-activity relationship (SAR) analysis and candidate prioritization in biomedical and clinical research pipelines.